All published articles of this journal are available on ScienceDirect.
Same-Site Deep Sclerectomy Re-Operation with Subconjunctival and Subscleral Collagen Matrix (Ologen™) and Minimal use of Mitomycin C
Abstract
Purpose:
This study aimed to evaluate the safety and efficacy of same-site Deep Sclerectomy (DS) re-operation with subconjunctival and subscleral collagen matrix (Ologen™) and minimal use of mitomycin C in cases of late failure blebs.
Methods:
A prospective evaluation of consecutive patients on which same-site DS was performed with subconjunctival and subscleral collagen matrix (Ologen™) and minimal use of mitomycin C; all performed at a single university institution. Intraocular pressure (IOP), number of glaucoma medications, postoperative interventions, postoperative complications and surgical success were registered at 24 hours, 1, 6 and 12 months after the surgery.
Results:
Twenty-five eyes of 25 patients were included in this study. Time from first DS was 55.08 ± 28.16 months. Mean preoperative IOP was 20.88 ± 6.06 mmHg and it was reduced to 16.64 ± 4.02 mmHg, at 12 months after re-operation (p 0,007). At this final visit, requirements for topical medications dropped from a mean of 2.2 ± 1.0 to 0.72 ± 1.0 per patient and no medications were required in 60% of patients. Success was 36 % in criteria A (IOP ≤15mmHg), 72% in criteria B (IOP ≤18 mmHg) and 80 % in criteria C (IOP ≤15 mmHg), 12 months after the second surgery.
Conclusion:
The same-site DS re-operation with subconjunctival and subscleral collagen matrix (Ologen®) and minimal use of mitomycin C is a safe and adequate technique for the reduction of IOP, especially when targeted IOP is not lower than 15mmHg.
1. INTRODUCTION
Glaucoma is one of the leading causes of blindness in the world and decreased Intraocular Pressure (IOP) has been proved to be a unique method to reduce disease progression. Deep Sclerectomy (DS) is a non-penetrating glaucoma surgery consisting of the removal of a deep scleral flap and the anterior wall of the Schlemm´s canal, that allows aqueous humor to flow through the anterior trabeculum into the newly created intrascleral space and through a newly formed subconjunctival bleb [1]. DS filtration blebs present a 10% to 20% late failure rate at 3 years after surgery, due to fibrosis and scarring of the subconjunctival or Tenon’s capsule tissue in most cases [2]. For this reason, adjunctive techniques are frequently used to increase surgical efficacy. These can include needling with mitomycin C (MMC), 5-fluorouracil (5-FU) or bevacizumab, insertion of spacer devices in the intrascleral space, supraciliary or the combination of both and, more recently, the insertion of a subscleral and subconjunctival collagen matrix [3, 4]. Ologen™ (Aeon Astron Europe BV. Leiden, The Netherlands) is a porcine skin-derived biodegradable collagen matrix implant used as an adjunct to trabeculectomy that has shown good results in several studies. It is a biodegradable tissue that prevents excessive scarring while acting as a reservoir for fibroblasts and helping to mechanically separate the conjunctiva and scleral surface, preventing adhesions between them [5, 6].
There is no current scientific consensus regarding the surgical approach to eyes with previously failed glaucoma surgery [7]. In non-penetrating glaucoma surgery, there is only one study that compared repeated DS versus aqueous tube shunts [8], but there are no publications on same-site repeated DS.
In this study, we report the surgical outcome of same-site DS with Ologen™ collagen matrix of failed non-penetrating glaucoma interventions.
2. MATERIALS AND METHODS
This is a prospective non-comparative study that includes patients with a late failure bleb who underwent same-site DS reoperation surgery between January 2015 and July 2017 at the Glaucoma Unit of Hospital Germans Trias i Pujol (Universitat Autònoma de Barcelona. Barcelona, Spain).
Regarding the first failed glaucoma surgery, a spacer device was implanted during the initial DS. These implants were a Esnoper V-2000® and Esnoper-Clip® (AJL Ophthalmics. Álava, Spain).
All the patients were informed of the nature of the study and signed a consent form approved by the Department of Ophthalmology. The study adhered to the tenets of the Declaration of Helsinki and was approved by the Institutional Review Board and Ethics Committee of the Hospital Universitari Germans Trias i Pujol. All the patients were over 18 years of age with a failed DS presenting uncontrollable open angle glaucoma, with an IOP considered excessive under maximal tolerable medical treatment. Patients who underwent same-site DS reoperation associated to phacoemulsification surgery were also included. In the preoperative assessment, patients underwent a complete examination including the best corrected visual acuity (BCVA) on a decimal scale, biomicroscopy, Goldmann tonometry, pachymetry (UP-1000®, Nidek, Japan) gonioscopy and retinal funduscopy. Follow-up visits were scheduled at 24 hours, 1, 6 and 12 months after the surgery. The number of anti-glaucomatous medications before the second DS and at the last follow-up was recorded (Table 1). Any further intervention needed was also recorded. This could include YAG-laser goniopuncture (Visulas® YAG III Combi, Carl Zeiss Meditec. Jena, Germany), YAG-laser iridoplasty (Visulas® YAG III Combi, Carl Zeiss Meditec. Dublin, USA) or needling with subconjunctival 5-FU.
Three levels of success criteria were defined:
Criteria A: Final IOP ≤15 mmHg and one of the following: ≥25% reduction of IOP or reduction of 1 anti-glaucomatous medication.
Criteria B: Final IOP ≤18 mmHg and one of the following: ≥20% reduction of IOP or reduction of 1 anti-glaucomatous medication.
Criteria C: Final IOP ≤21 mmHg and one of the following: ≥20% reduction of IOP or reduction of 1 anti-glaucomatous medication.
2.1. Surgical Technique
Before the surgery, all the patients underwent a gonioscopy to assess the transparency of the TDM and anterior segment optical coherence tomography (AS-OCT) (Visante OCT, Carl Zeiss Meditec. Jena, Germany) of the intrascleral bleb to confirm the viability of the membrane. All surgical interventions were performed by the same experienced surgeon (JLA). A superior 4/0 nylon traction suture was passed through the superior cornea and a fornix-based conjunctival flap was dissected, followed by cauterization of bleeding vessels. Conjunctiva was dissected and 0.02% MMC was applied for one minute and then profusely irrigated with saline solution. The scleral flap was re-dissected exposing the implant which was reviewed and repositioned if necessary, otherwise, it remained in its position without any modification. Aqueous drainage through the TDM was searched before the scleral flap was closed with two loose sutures. The Ologen™ collagen matrix was placed one half under the superficial scleral flap while the other half rested under the conjunctiva, communicating both the spaces (Figs. 1 and 2). The conjunctiva was then sutured with 10/0 nylon. Eyes scheduled for cataract surgery underwent a lens extraction before the same-site DS. It included standard phacoemulsification, intraocular lens insertion in the capsular bag (Akreos MICS MI60L, Bausch and Lomb. Rochester, USA) and a corneal suture. Postoperative treatment consisted of topical ofloxacin (Alcon Cusí SA. El Masnou, Barcelona, Spain) three times a day for one week and dexametasone 1mgr/mL (Alcon Cusí SA, El Masnou, Barcelona, Spain) six times a day, tapering over six weeks. Repeated goniopuncture was performed when percolation of aqueous humor was considered insufficient. In the same way, needling procedure was performed under topical anesthesia using a 27-gauge needle and 5-FU was injected subconjunctivally when it was considered necessary by the investigators.
| Num patients | 25 |
| Sex (man/female) | 11/14 |
| Mean age (years ± SD) | 71 +/- 10,3 |
| Eye (right/left) | 10/15 |
| Type of glaucoma | |
| POAG | 15 |
| PEXG | 6 |
| PG | 1 |
| NTG | 2 |
| JG | 1 |
| Type of implant in previous surgery | |
| Esnoper V-2000 intrascleral | 9 |
| Esnoper V-2000 supraciliary | 11 |
| Esnoper Clip | 5 |
| Type of surgery | |
| Bleb revision with ologen | 14 |
| Bleb revision with ologen plus Phacoemulsification | 11 |
| Time between surgeries (months) | 55.08 +/- 28.16 |
| VA preop (Snellen)* | 0.6 +/- 0.27 |
| VA 1 year (Snellen)* | 0.6 +/- 0.27 |
| IOP preop (mmHg)* | 20.88 +/- 6.05 |
| IOP 1 year (mmHg)* | 16.64 +/- 4.02 |
| Preop Med* | 2.2 +/- 1.0 |
| 12 months Med* | 0.72 +/- 1.0 |
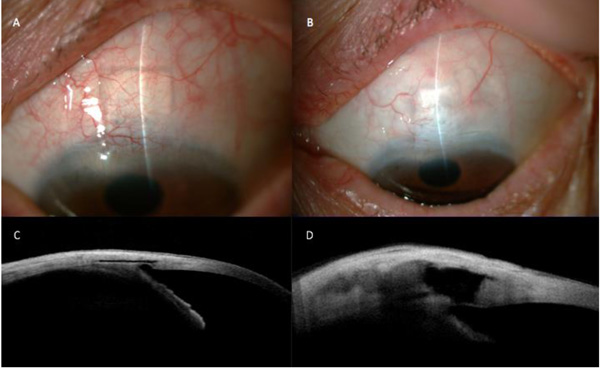

3. RESULTS
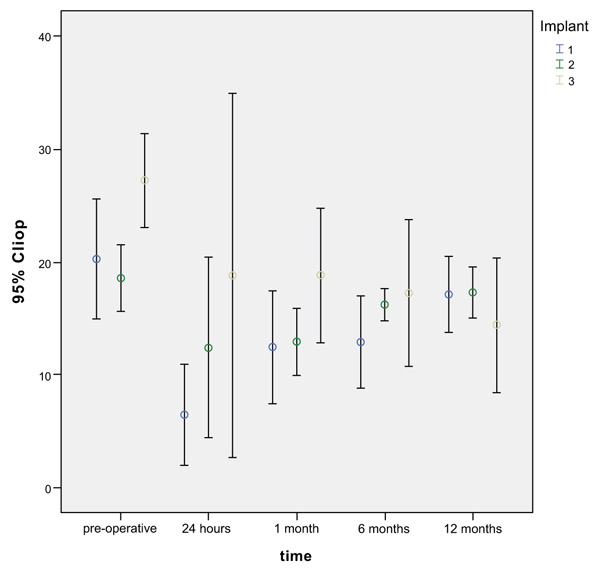
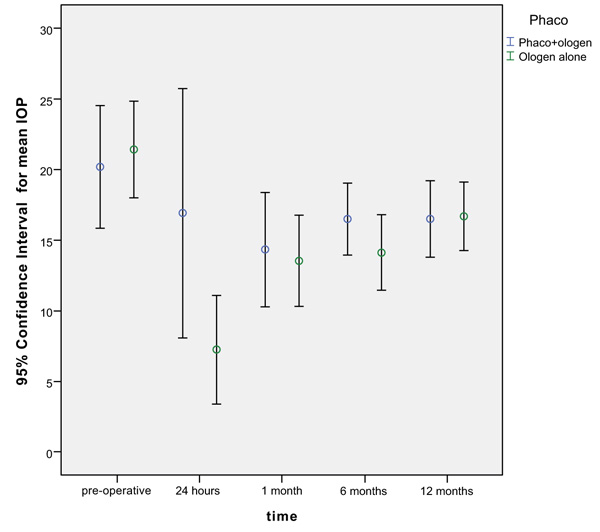
Twenty-five eyes of 25 patients were prospectively included in this study. All the patients were followed for 12 months and data was recorded. The time from the first surgery was 55.0 ± 28.2 months. Other demographic data are shown in Table 1. Mean IOP was reduced from 20.88 ± 6.06 mmHg and 2.2 ± 1.0 anti-glaucomatous medications to a postoperative IOP of 16.64 ± 4.02 and 0.72 ± 1.0 anti-glaucomatous medications at 12 months (20.3% reduction) (Table 2) and Fig. (3). The reduction in IOP and in the mean number of medications was statistically significant. At the 12 months visit, 60% of the patients did not use any antihypertensive medication to control the IOP. Two patients required needle revision with 5-FU (8%), one patient received a subconjunctival injection of bevacizumab (4%) and 2 cases a goniopuncture (8%). Taking into consideration which implant was used in the original DS, there were no statistically significant differences in mean IOP reduction at 24 hours (p = 0.288) and 1 month (p=0.645). The differences between implants were near to statistical significance at 6 months (p=0.063) and strongly significant at the 12 months visit (p = 0.004) (Fig. 4). The uveoscleral implant group (Group 3) differed significantly from the supraciliary implant (Group 1) (p = 0.007) and the intrascleral implant (Group 2) (p = 0.001), while groups 1 and 2 were not significantly different (Fig. 4). Mean IOP reduction at 12 months was 3.11 ± 5.58 mmHg in group 1, 1.27 ± 5.23 mmHg in group 2 and 12.80 ± 7.29 mmHg in Group 3. In combined procedures with cataract surgery compared with DS with Ologen™ alone (Fig. 5), IOP reduction was statistically significant only at 24 hours after the surgery (p=0.008), but not at any the further visit (Table 3) , with a higher IOP in the combined procedure.
| Time | IOP | p-value (compared with pre-operative) | Number of medications | p-value (compared with pre-operative) |
|---|---|---|---|---|
| pre-operative | 20.88 +/- 6.06 (12-36) | 2,2 +/- 1,00 | ||
| 24 hours | 11.52 +/- 10.93 (2-38) | 0,0002 | 0,00 +/- 0,00 | <0,0001 |
| 1 month | 13.92 +/- 5.67 (4-24) | <0,0001 | 0,16 +/- 0,47 | <0,0001 |
| 6 months | 15.20 +/- 4.36 (4-24) | 0,0003 | 0,48 +/- 082 | <0,0001 |
| 12 months | 16.64 +/- 4.03 (10-24) | 0,007 | 0,72 +/- 1,06 | 0,0003 |

| Time | re-DS (IOP) | re-DS+Phaco (IOP) | p-value (between 2 groups) |
|---|---|---|---|
| pre-operative | 21.43 +/- 5.89 | 20.18 +/- 6.48 | 0,643 |
| 24 hours | 7.29 +/- 6.63 | 16.91 +/- 13.13 | 0,008 |
| 1 month | 13.57 +/- 5.61 | 14.36 +/- 5.99 | 0,424 |
| 6 months | 14.14 +/- 4.61 | 16,55 +/- 3,80 | 0,182 |
| 12 months | 16.71 +/- 4.20 | 16.55 +/- 4.01 | 0,716 |
According to success criteria A, B and C at 12 months, Kaplan-Meier survival curves Fig. (6) and Table 4 show a complete success of 36 % in criteria A, a complete success of 72% in criteria B and a complete success of 80 % in criteria C. Comparing the type of implant and type of procedure, the survival curves were not statistically significantly different in any of the criteria.
Time from original surgery to reoperation and its potential influence on final IOP were also analysed. Regression coefficients of months since first surgery were -0.047 at 24 hours, -0.0168 at 1 month, 0.03 at 6 months and 0.082 for 12 months (all of them with no statistically significance). Statistical evidence for the observation that it has been a long time since the first surgery induces an unfavourable outcome is weak.
| Month |
Success according to criterion A |
Success according to criterion B |
Success according to criterion C |
|---|---|---|---|
| 1 | 0.64 | 0.84 | 0.92 |
| 6 | 0.48 | 0.84 | 0.92 |
| 12 | 0.36 | 0.72 | 0.8 |

The association between success and months since first surgery was also investigated with a Cox regression model. The relative risk for criteria A was estimated as 0.85 per year (95% CI: 0.65 – 1.11), meaning that for each year since the first surgery the probability of success is worse. This finding was not significantly different from 1 (p=0.23).
Postoperative complications were low. We found 2 cases of Seidel phenomena at 24 hours (8%), two of athalamia (8%), 3 of hyphema (12%), all resolved within the first week, and 1 case of iris synechia or incarceration (4%) that resolved with laser iridoplasty.
4. DISCUSSION
This is the first study comparing the efficacy and safety of same-site DS with subconjunctival and intrascleral Ologen™ collagen matrix and minimal use of MMC.
A conjunctival bleb is considered as “failed” when it is completely flattened with an elevated IOP and the need for glaucoma medications. There are two main surgical options to solve this failure: performing a new DS in a different site (repeated DS) or trying to repermeabilize the existing DS (same-site DS). Few studies have reported the long term outcomes of repeated glaucoma surgery on a previously failed one and in all of them, trabeculectomy was done [7, 9-14]. There is only one study comparing surgical outcomes of reoperation with Baerveldt shunt implant versus different site DS. [8] Our study is the first publication showing results in same-site DS: its surgical technique and the first year outcome.
Bergin et al. [8] who compared different site DS versus Baerveldt tube implantation, found a mean IOP reduction in the DS group from 19.9 ± 6.1 mmHg with 2.4 ± 1.0 medications to 14.1 ± 5.2 mmHg with 0.8 ± 1.1 medications at 12 months (a 29.1% mean IOP reduction). Using the Kaplan–Meier, failure was estimated by the authors at 30% in the DS group, at year 1. Hirungpatravong et al. [12] Meyer et al. [10] and Rodriguez et al. [14] described the results after same-site trabeculectomy. Comparing our results using similar criteria, Hirungpatravong et al. [12] had a success of 69.7% for criteria A (final IOP ≤18 mmHg) and 60.9% for criteria B (final IOP ≤15 mmHg) at 1 year, Meyer et al [10] 37.5% and 32.1% respectively at 2 years and Rodriguez et al. [14] a success rate of 53.7% in ≤17 mmHg target and 51.2% at t ≤14 mm Hg target at 5 years. El-Saied et al. [15] performed repeated trabeculectomy with Ologen™ collagen matrix on a previously failed one in secondary glaucomas, showing a mean IOP reduction of 55% at 1 year.
Wlaz et al. [16], performed phacotrabeculectomy comparing the efficacy of collagen matrix versus MMC, he reported IOP reduction from 26.4 ± 6.1 to 13.7 ± 3.8 (52%) in the Ologen™ group and 23.4 ± 3.6 to 13.3 ± 2.8 (56%) in the MMC group, with no significant differences in the cumulative success rates by Kaplan–Meier analysis, 92,6% Ologen™ group and 92,3% MMC group, respectively. Castejón et al. [17], analyzed the results achieved in lowering intraocular pressure (IOP) after trabeculectomy or phacotrabeculectomy with low-dose MMC using Ologen™, he reported statistically significant differences in IOP reduction only in the phacotrabeculectomy group + Ologen™ at 1 and 2 years follow up (p 0.0008 and 0.02, respectively). Both concluding that Ologen™ in adjunct to MMC in filtering surgery improves postoperative IOP results over the long term.
Differences with our results could be explained by the differences in the surgical technique. The success rate at 12 months of 36 % in criteria A, 72 % in criteria B, and 80 % in criteria C in the Kaplan-Meier survival curves suggests that the technique of same-site DS could favor patients in whom target IOP is between 15 and 21 mmHg but not patients with a stricter IOP target (≤ 15 mmHg). In these cases, it could be convenient to evaluate other surgical options such as drainage devices, different site DS or trabeculectomy.
We could not find a statistically significant IOP difference between same-site DS with or without phacoemulsification. Same-site DS technique could be considered when phacoemulsification must be done after late failure bleb and even more if we want to decrease the number of medications.
The Esnoper V-2000® is a single triangular shaped form plate that can be placed in the supraciliary space (Group 1) or the intrascleral space (Group 2), while Esnoper-Clip® is a uveoscleral implant (Group 3) designed with 2 footplates, one placed in the intrascleral space and the other in the supraciliary space. [13]
Comparing the three implants used in the first intervention, IOP reduction was achieved in all three implant groups during the first year after the second DS, but these results were better when a uveoscleral implant was used in the first DS, compared to the supraciliary and intrascleral implants.
In our study, a longer time interval between the first and second same-site DS was not associated with an increased failure rate. This finding differs from what has been published by Hirungpatravong et al [12] who reported that an interval of less than 3 years between the first and the second trabeculectomy is associated with a significantly higher rate of failure.
We did not find any severe surgical complications or any case of hypotensive maculopathy in our study group. This may be relevant when reintervention surgical options have to be considered in cases of late bleb failure. Glaucoma surgeons should be aware of iris incarceration in the postoperative period and even more in combination with goniopuncture. To avoid this complication, we recommend the use of topical pilocarpine during the early postoperative stage or a YAG-laser iridoplasty in selected cases. Same-site DS also preserves more the conjunctiva and reduces the risk of surgical complications related to trabeculectomy or drainage devices.
CONCLUSION
Same-site DS with subconjunctival and subscleral collagen matrix (Ologen™) and minimal use of MMC is an easy, safe and adequate technique that preserves normal conjunctiva and significantly decreases IOP in cases of late bleb failure. However, in eyes that require a very low postoperative IOP, the use of drainage devices or different site DS procedure should be considered.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The study was approved by the Institutional Review Board and Ethics Committee of the Hospital Universitari Germans Trias i Pujol.
HUMAN AND ANIMAL RIGHTS
No Animals were used in this research. All human research procedures followed were in accordance with the ethical standards of the committee responsible for human experimentation (institutional and national), and with the Helsinki Declaration of 1975, as revised in 2013.
CONSENT FOR PUBLICATION
All the patients were informed of the nature of the study and signed a consent form approved by the Department of Ophthalmology.
FUNDING
None.
CONFLICT OF INTEREST
The authors declares no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Declared none.


