All published articles of this journal are available on ScienceDirect.
Testing Spatial Detection and Light Sensitivity in Homonymous Hemiano-pia by Rarebit and Conventional Automated Perimetry
Abstract
Purpose of the study was to compare Rarebit Perimetry (RP) with Standard Automatic Perimetry (SAP) in patients with ischemic retrochiasmal lesions. Seventeen patients with homonymous hemianopia underwent SAP and RP. Correlation between the two techniques was evaluated, congruent defects were quantified in the spared hemifield and residual vision areas were investigated in the blind hemifield. A strong correlation was found between SAP and RP. In the spared hemifield, RP detected congruent defects in a higher percentage of cases. Residual detection function has been found at RP in 69,2 % of perimetrically blind visual field quadrants as assessed by SAP. In conclusion, RP appears to be useful to assess visual function in hemianopic patients. As compared to SAP, it seems able to detect small congruent defects in a higher percentage of cases. Finally, a certain residual function seems to be present at RP in the blind regions of the visual field, however, for this finding further demonstrations are required.
INTRODUCTION
Visual field defects are a main consequence of cerebrovascular accidents and cerebral tumours due to the wide representation of the visual pathway in the central nervous system [1].
It has been estimated that in about 20-30% of strokes visual system is involved to a certain degree [2,3].
Consequently, typical visual field defects that are hemianopia or quadrantanopia are frequently found in patients using Standard Automated Perimetry (SAP). In the last few years, although attempts have been made to find a more effective perimetric test in evaluating such neurophthalmological disorders, conventional perimetry remains the gold standard. In fact, neither Frequency Doubling Technology (FDT) [4-8], nor objective perimetry as multifocal VEP [9] or pupillometry [10] have proved to be more sensitive. Only Short Wavelength Automated Perimetry (SWAP) seems to be more sensitive than SAP in demyelinating or vascular diseases [11-13] but, as already stated [14], SWAP usefulness is strongly limited.
It is important to stress that hemianopic defects as tested by SAP are referred to as light sensitivity. Nonetheless, there is evidence that other visual functions [15-21] could result in a different degree or extent of damage in the affected hemifield or after geniculostriate differentiation.
Rarebit Perimetry (RP) is a new perimetric test developed by Frisen [22-24] which seems to be promising in the detection of visual field damage in patients with neurological diseases. It differs from other perimetric techniques as it measures the frequency in detecting minimum informational stimuli across the visual field, that is microdots displayed for a brief time and at a suprathreshold level. Under this basis, rather than testing light sensibility we can assume it measures a different function that is visual detection over space [22, 25, 26].
In this study spatial visual detection has been evaluated in patients affected by homonymous hemianopia/quadrantanopia by means of RP. Quadrantanopias were considered as incomplete hemianopic defects, so both types of patterns were combined and analysed as a single group. Results have been correlated with SAP in order to estimate if RP can be more informative in discovering minimal defects in the spared hemifield and in assessing functional differences in the compromised hemifield.
MATERIALS AND METHODS
The rarebit technique has been described in detail in a previous article by Frisén [22]. It aims to evaluate the neuroretinal matrix looking for gaps in receptive field spatial continuity. Rather than spots of light on a white background, as it is used in conventional perimetry, visual stimuli employed are two high contrast white microdots (dot size: 50% of the minimum angle of resolution, MAR) on a dark background of a PC liquid crystal screen. Dot contrast does not change during the examination while dot size increases with eccentricity, so as to keep its MAR constant in every locus in the visual field. Patients are presented two stimuli separated by 4° are displayed simultaneously at random positions for a brief time (200 ms).
Examination was performed in monocular conditions. The patient is required to look at a fixation cross and to indicate if no dots, one dot or both dots are perceived by not clicking, clicking or double-clicking a mouse button. Under these conditions and according to the test program, we have chosen 24 rectangular areas (6°x8°wide in the centre and 6°x14° wide in periphery) covering an overall visual field extended 30° horizontally and 20°vertically from the fixation point. In each area, dot detection is repeatedly evaluated n- times (or passes), until the technician interrupts the test. For our experiments we adopted ten passes which strike a balance between high degree accuracy and examination duration.
The main characteristic of rarebit technique is its probabilistic approach. Rather than thresholding visual function, the test expresses functional integrity of visual field in every locus as a percentage of hit rates over the total number of presented stimuli.
The program provides a global index, Mean Hit Rate, expressed as percentage (MHR, ± SD) and obtained by averaging single results from every locus except the two areas next to the blind spot. The lower normal limit for MHR has been set at 92.7% [23]. In SAP correspondent index it may be regarded as Mean Sensitivity (MS) 17 patients (12 males, 5 females) suffering from homonymous hemianopia or quadrantanopia as diagnosed at SAP and resulting from post-stroke cerebral lesion as documented at CT and MNR were recruited. Of these patients, 10 (58,8%) had an hemianopia and 7 (41,2%) a quadrantanopia. The time of onset of neurological symptoms varied from 2 to 117 months (Table 1).
Demographic and Anamnestic Data of Recruited Patients. Subjects 15, 16 and 17 were Excluded for Low Reliability of their Examinations
| Patient | Sex | Age (y) | Months Since Onset | Refraction (sfer. equival.) (R / L) | Localization | Perim.Pattern |
|---|---|---|---|---|---|---|
| 1 | f | 74 | 2 | 4,00/4,25 | temporal-occipital R | LEFT HEMI |
| 2 | m | 52 | 84 | -2,00/-3,00 | frontal+parietal+temporal R | LEFT HEMI |
| 3 | f | 72 | 11 | 1,25/1,25 | occipital R | LEFT HEMI |
| 4 | m | 64 | 25 | 1,00/0,75 | occipital L | RIGHT HEMI |
| 5 | m | 56 | 20 | 3,25/1,00 | occipital R | LEFT HEMI |
| 6 | f | 68 | 9 | 1,25/1,00 | frontal+par+tem+occipital R | LEFT HEMI |
| 7 | m | 34 | 60 | -1,75/-1,75 | occipital R | LEFT HEMI |
| 8 | m | 56 | 117 | -1,50/-2,00 | vertebro-basilar R | LEFT HEMI |
| 9 | m | 75 | 32 | -2,00/-1,50 | temporal-occipital R | LEFT HEMI |
| 10 | m | 80 | 13 | 0,75/0,50 | occipital. R | LEFT HEMI |
| 11 | m | 68 | 2 | -0,50/1,00 | frontal+temporal R | LEFT HEMI |
| 12 | m | 70 | 40 | -1,25/-1,25 | temporal L | RIGHT SUP QUAD |
| 13 | f | 63 | 50 | 1,50/1,50 | occipital L | RIGHT SUP QUAD |
| 14 | f | 39 | 11 | -3,50/-5,00 | Not specific | LEFT INF QUAD |
| 15 | m | 66 | 4 | 1,50/1,00 | frontal+temporal L | RIGHT INF QUAD |
| 16 | m | 51 | 18 | -1,25/-1,25 | parietal+occipital L | RIGHT INF QUAD |
| 17 | m | 71 | 14 | 2,25/2,25 | occipital R | LEFT SUP QUAD |
| MEAN | 62,3 | 30,1 | ||||
| SD | 12,7 | 31,7 | ||||
| RANGE | 34-80 | 2-117 | ||||
Congruence Levels Analysis in the Spared Hemifield. In Brackets is SD
| SAP | RP | p | |
|---|---|---|---|
| CLSAP4; CLRP10 (locations %) | 12,85 (13,89) | 33,57 (18,65) | 0,003 |
| CLSAP2; CLRP5 (locations %) | 9,34 (9,52) | 19,29 (11,41) | 0,017 |
| hemifields with congruent defects for CLSAP4 and CLRP10 (%) | 78,5 | 92,80 | ns |
| hemifields with congruent defects for CLSAP2 and CLRP5 (%) | 78,5 | 92,80 | ns |
P-Values are Given by Fisher' Exact Test.
According to Wall et al. [4], a visual field defect was considered as hemianopic (or quadrantanopic) if the whole hemifield (or quadrant) was depressed with a sharp separation along the vertical meridian and without any other abnormal test location that would obscure the homonymous pattern.
Mean age was 62,3 years (±12,7, range 34-80). Exclusion criteria were best corrected visual acuity (BCVA)< 0.7, ametropia>±3 diopters, ophthalmological diseases, intraocular pressure higher than 18 mmHg (G) or inheritance for glaucoma, strabismus, amblyopia, diabetes, systemic therapies able to induce visual field defects, cognitive disability and poor cooperation. Every patient showed stable foveal fixation as tested by Cuppers ’ visuscope.
The Neuro-Ophthalmology Service of the Opthalmology Department of Gradenigo Hospital, Turin, recruited all subjects.
As a control group, 14 normal subjects (mean age=57 years, ±10,85, range 34-70) were enrolled from the staff of the hospital. Exclusion criteria were the same as those of patients. subjects with systemic hypertension or inheritance for cerebral vascular diseases were excluded. All control subjects had normal or corrected to normal visual acuity.
After obtaining informed consent, subjects underwent a complete ophthalmological examination (including slit lamp examination, retinoscopy, tonometry, visual acuity measurement, pupillary reflexes and ocular movement evaluation). Conventional automated perimetry was performed by means of an Octopus 1-2-3 perimeter (Haag-Streit, Bern, Switzerland), program 32x, which analyses central 30° visual field (76 locations on a 6°-spaced grid) with a Goldmann size III stimulus (0,43° wide).
Rarebit perimetry (version 4) was performed first at a distance of 50 cm to test peripheral loci (”outer” modality), then at 1 meter to test central loci (“inner” modality). “Outer” and “inner” modalities are not modification of the original methodology, which automatically provides to adjust the size of the targets and their separations appropriate to the two viewing distances employed.All patients were acquainted in conventional perimetry while rarebit examination was preceded by a short training course (10 minutes) to increase confidence with the new technique. Calibration of the instrument was performed before every trial.
Sessions took place in a dimmed room (average illumination: 1 lux). The patients head was kept fixed by a chin rest at a distance of 0,5 meters to test the 20 peripheral areas and later at 1 meter to evaluate the remaining 4 inner locations. Adequate lens was used for the examination distance and eventually to correct refractive defects.
The same operator, skilled in psychophysical procedures, tested eyes in random order.
Reliable criteria for conventional perimetry were: reliability factor (RF) < 25 % and false positive answer less than 10%, while for Rarebit: error statistic value ≤ 4 (for 10 passes).
The study considered:
- for SAP: MS, MS in the affected and spared hemifield. To investigate congruous defects in the spared hemifield, the percentage of significant depressed points (or clusters) (p < 5%) with corresponding defect in the spared hemifield of the fellow eye were calculated in the Comparison map.
In this case the adopted arbitrary criterion was:
- Exact spatial correspondence (that is absolute congruence) of the two points (or clusters) in the two eyes with a difference ≤ 4 dB, or
- The same but with a difference ≤ 2 dB.
These specific tolerance levels would correspond to the upper limit of light threshold fluctuation as referred in normal subjects [27,28].
We will refer to these values as SAP Congruence Levels (CLSAP4, CLSAP2).
To minimize bias due to fixation loss, the six points around the blind spot were not considered.
- For RP: MHR (excluding the two areas closest to the blind spot), MHR in the affected and spared hemifield. To investigate congruous defects in the spared hemifield the percentage of areas with Miss Rate ≥5% with corresponding defective areas in the spared hemifield of the fellow eye has been calculated in the RP Miss Rate map.
In this case the adopted arbitrary criterion was:
- Exact spatial correspondence of the two areas in the two eyes with a Miss Rate difference ≤ 10%, or
- The same but with a Miss Rate difference ≤ 5%.
These values are close to the upper limit of Short Term and Long Term Fluctuation in Rarebit Perimetry in a global and point-to point analysis [29].
We will refer to these values as RP Congruence Levels (CLRP10, CLRP5 ).
In these cases the two areas corresponding to the blind spot were not considered in the spared hemifield.
To assess functional differences between light sensitivity and spatial detection in the affected hemifield, areas with residual function detected at RP and MHR was evaluated in each perimetrically blind quadrant as tested by SAP. In this case quadrantanopias have been excluded and only hemianopias (10 patients) were examined.
A quadrant was considered as perimetrically blind if one of these two conditions were satisfied at SAP:
- Absolute scotoma in each of the nineteen locations of the quadrant, or
- Deep depression (sensibility ≤ 5 dB) involving ≤ 4 locations along the edge of the total length of vertical meridian with abolished sensitivity in the remaining points of the edge and in the rest of the affected quadrant. In this case, the residual level of sensibility along the vertical meridian has been ascribed to fixation shift.
However, to avoid any influence by fixation instability or fixation losses in hemianopic visual fields, in the rarebit map the two indexes were evaluated only on the three more peripheral areas (located from 10° to 30° of eccentricity), excluding the three remaining locations nearest to the vertical meridian.
When tested by RP, an area was regarded as having a residual function if its Hit Rate value was ≥ 15% in a first estimation or as a second paradigm, ≥10%. For these two cases, two different analyses were done.
Correlation between MS and MHR in the whole field and between hemifields was analysed by Pearson’s correlation analysis. Kolmogorow-Smirnow test was used to test for normality. Paired t-test was used to compare examination time and differences between patients and control subjects. Mann-Withney test was employed to analyse differences between results in both examinations. Statistical significance level was set for p value ≤ 0,05.
After mean values, in brackets is given standard deviation.
We certify that the research followed the tenets of the Declaration of Helsinki, that informed consent was obtained from the subjects after explanation of the nature and possible consequences of the study and that all applicable institutional and governmental regulations concerning the ethical use of human volunteers were followed.
RESULTS
Of the 17 recruited patients, 14 completed the experiment while 3 were excluded for the high rate of errors in one or both tests. None of the 14 normal subjects demonstrated any visual field defect at SAP.
Mean MS in the control group and in the group of patients was 27,7 dB (±0,81) and 14,7 dB (±3,60), while MHR was 96,8 % (±1,75%) and 52,2 % (±12,30%) (Difference: p≤0, 01 in both cases). In both groups RP examination time was significantly lower compared to SAP examination time performed by Octopus 1-2-3 (in controls: RP: 568,2 sec (±33,96), SAP: 1211,7 sec (±55,82); in patients: RP: 648,79 sec (±56,91), SAP: 1138,54 sec (±128,62), p≤0, 01). Topographical correspondence of damage was very high in the two maps (Fig. 1).

Topographical correspondence between SAP and RP in a 64-year old patient with right homonymous hemianopia.
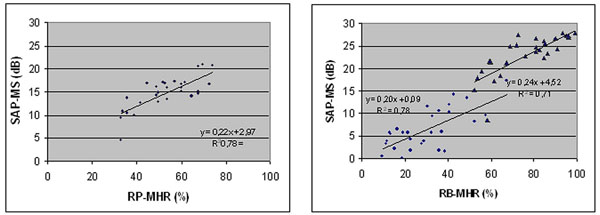
Correlation between Standard Automated Perimetry Mean Sensitivity (SAP-MS) and Rarebit Perimetry Mean Hit Rate (RP-MHR) in patients affected by homonimous hemianopia and quadrantanopia. Left: whole field; Right: spared (circles) and affected (triangles) hemi-fields.
In Fig. (2) is shown SAP/RP correlation analysis. Regarding the whole field significant correlation was found between SAP MS and RP MHR (r= 0,78, p < 0,01) (Fig. 2, left).
Correlation coefficient between RP MHR and SAP MS in the spared hemifield resulted to be the same as that related to the whole perimetric map (r= 0,78, p < 0,01)), while a slightly lower correlation was found in the affected hemifield (r=0,71, p < 0,01) (Fig. 2, right).
To quantify the congruence level of damage in the spared hemifields of both eyes, corresponding defective locations as a percentage have been calculated. It has been made for two values of defect in SAP and RP as stated previously. As shown in Table 2, percentages of defective points in the spared hemifield were higher with RP than with SAP, for both congruence levels. Compared to SAP, RP was able to find congruent defects in a higher number of patients, irrespective of the CL value. Considering each technique, there were no differences in the number of defective points/areas detected using the two CLs (p>0,05).
To investigate residual visual function in the perimetrically blind region of the visual field the number of areas and MHR for each blind quadrant was evaluated, as specified in the methods section. Of the 40 quadrants obtained by SAP maps of our 10 hemianopic patients, 13 quadrants (32,5 %) were found to be perimetrically blind as tested by SAP according to our criteria. In 9 of these (69,2%) some residual function was found when tested by RP with MHR cut-off set ≥ 10% and in 8 (61,5%) with MHR cut-off set ≥ 15%.
In the perimetrically blind quadrants, the total number of residual areas was 21 (1,6 area per quadrant) (cut-off ≥ 10%) and 13 (1 area per quadrant) (cut-off ≥ 15%).
No correlation was found between residual MHR and onset time of accident (Spearman rs =-0,05, p > 0,05) or age of patients (Spearman rs=-0,15, p >0,05).
DISCUSSION
It is well known that one of the major shortcomings of perimetry testing is its duration. An alternative technique to Standard Automated Perimetry, able to test rapidly visual field of patients affected by cerebral stroke is convenient, particularly considering their peculiar conditions, being often unable to perform a tiring full threshold examination. The Rarebit Perimetry has been demonstrated to be useful in ophthalmological practice [22-24, 30-36]. A recent study [37] has suggested it to be as effective as Humphrey standard automated perimetry in detecting homonymous hemianopia.
According to our results, rarebit technique could be a valid alternative in hemianopic patients considering its good correlation to Octopus Standard Automated Perimetry (taken as gold standard) and its short duration. Test-retest variability (in normal subjects) seems not to be greater than that found in SAP as found by Frisén [22] and Corallo [29].
Once the presence of hemianopia has been assessed, attention in ophthalmological follow-up is focused in particular on the spared hemifield to survey the onset of new defects. In this respect, results we obtained suggest a higher sensitivity in detecting localized congruent defects for RP as compared to conventional perimetry. As already suggested [22], a possible explanation can rely on the strong reduction of informational content in the RP target, as compared to that used in conventional perimetry and in the lower light scattering from microdot stimuli so that defective areas in the spared region were not affected by stimuli projected to a neighbouring location.
It has been underlined in a previous study the problem of scattered abnormal depressed regions that may confound the typical hemianopic pattern [4]. Due to the microdot stimulation, a localized reduction in MHR can result as a consequence of physiological angioscotoma, without reflecting a real defect [38, 39].
In the blind regions of impaired visual fields, there is wide psychophysical (and electroretinographical [40]) evidence of the persistence of a little amount of visual sensibility. In particular some functions as motion perception [16-19,41], spatial vision detection [1, 20] or color [18,21] and orientation discrimination [21] result to be partly spared or less affected than light sensibility. The case of the patient who is aware of his residual visual perception (i.e. a different condition than the so-called blindsight [41]) has been ascribed to activity of spared occipital cortical cells [17,42] or optic fibres of the geniculo-striate visual pathway [43].
Indeed, the finding at RP of residual visual function in the perimetrically blind region (as assessed by SAP) supports this eventuality.
Caution must be used in this conclusion, for the possibility of a light scattering effect inducing an illusory perception in the blind region or, more probably, of a fixation shift affecting spatial localization of the perceived target [44-46]. Indeed a scattering effect seems to be less effective than in conventional perimetry due to the very small size of the RP stimuli. In recruited patients neither eccentric fixation nor macular plitting at SAP was found and fixation stability would be due to foveal sparing, covering a field extension of about 1,5 deg [46-48]. On the other hand, in rarebit perimetry it is impossible to detect and neutralize presentations occurring during a shift of fixation since a way for controlling this variable is not available. For this reason residual function analysis was performed only in those locations more than 10 deg apart from the midline, though this way a great amount of information across the visual field would inevitably be lost.
About this point, however, further confirmations are required, using at SAP a wider stimulus or an higher resolution grid (1 deg wide).
CONCLUSION
In conclusion, rarebit perimetry is a rapid and suitable perimetric technique in patients suffering from post-stroke visual field impairment. It appears to be more sensitive than SAP in detecting congruent defects in the spared hemifield. For these reasons it seems valid either as a screening or a follow-up test in patients with hemianopic and quadrantanopic defects. Moreover, if our data are confirmed by further research, RP could be employed to explore residual visual function in the blind regions of the visual field.


