All published articles of this journal are available on ScienceDirect.
The Effect of Intravitreal Azithromycin on the Albino Newborn Rabbit Retina
Abstract
Purpose:
To evaluate the effect of intravitreal azithromycin on the retina in a newborn rabbit model.
Methods:
Twelve, two-week old New Zealand albino rabbits were divided into two groups (six in each). The right eyes of six rabbits received 0.75 mg (0.05 mL) azithromycin and the right eyes of the remaining six rabbits 1.5 mg (0.1 mL) azithromycin intravitreally. Left eyes were served as the control and received the same volume of saline. All eyes were enucleated at the third postinjection week. Retinal histology was examined by light microscopy. Apoptosis of the retinal cells was further evaluated by immunohistochemical staining for caspase-3 and in situ terminal deoxynucleotidyl transferase-mediated biotin-deoxyuridine triphosphate nick-end labeling (TUNEL) of DNA fragments.
Results:
Light microscopy demonstrated no retinal abnormalities in all eyes. However, retinal nuclear DNA fragmentation was evident in both study groups (33.6% with 1.5 mg and 21.4% with 0.75 mg azithromycin) with the TUNEL method. TUNEL staining ratio was statistically higher only in the second group treated with 1.5 mg azithromycin when compared to the control group (p=0.01 Mann Whitney U test). The ratio of caspase-3 positive cells in the two study groups was 21.5% and 20.2%, respectively. Caspase-3 staining ratio was statistically higher in both study groups when compared to the control eyes (p=0.00, p=0.00 respectively). The difference of TUNEL staining ratio between the two study groups was statistically significant (p=0.028), but there were no statistically significant differences in the two study groups by caspase-3 staining (p=0.247).
Conclusion:
In newborn rabbits, intravitreal azithromycin injection resulted in an apoptotic activity in the photoreceptor, bipolar and ganglion cells. Immunohistochemical analysis suggested that doses of 0.75 mg and 1.5 mg azithromycin, administered intravitreally might be toxic to the newborn rabbit retina.
INTRODUCTION
Azithromycin is a second generation macrolide characterized by a broad antibacterial spectrum and acts as an bacteriostatic agent [1]. Its capability of intracellular penetration creates a certain advantage in the treatment of many intracellular organisms such as Chlamydia, Legionella, Mycobacteria, Bartonella and Toxoplasma species [2-6]. Azithromycin (Azyter 1.5%; Thea Laboratories, France) was marketed commercially in 2008 as an eye drop and nowadays is mostly preferred in children due to its short treatment course and reduced drop frequency for the treatment of bacterial conjunctivitis [7, 8]. It is available in a single-dose preservative free form commercially. However as it is a lipophilic compound and not stable in aqueous solution it is dissolved in a medium chain thrygliceride hydrophobic excipient namely Miglyol. Although topical azithromycin is widely used it has not been administered intravitreally and thereby its effect on the retina has not been reported. Intravitreal injection of azithromycin can at least find a place in the treatment of toxoplasma retinochoroiditis theoretically. In the present study, we investigated the effect of intravitreal azithromycin on the retina in a newborn rabbit model.
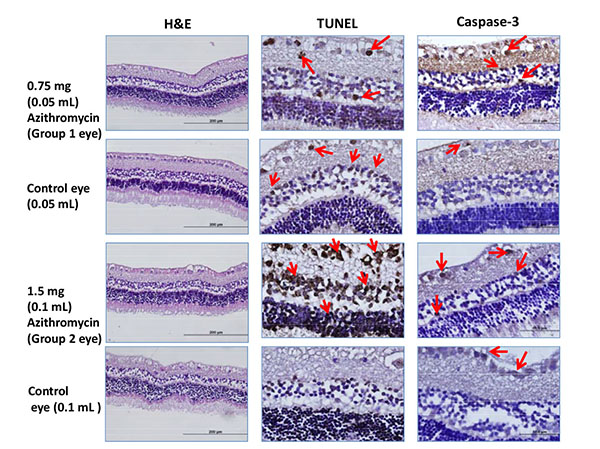
Examples of Hematoxyline eosine(H&E),TUNEL and caspase staining for each group. TUNEL and caspase positive cells are more in numbers than the controls.Arrows point out the brown stained TUNEL and caspase-3 positive sensorial layer retinal cells.
MATERIALS AND METHODS
Animals
Twelve newborn New Zealand albino rabbits weighing between 0.3 and 0.6 kg were used for this study. All experimental protocol was approved by the Animal Care and Use Committee of Dokuz Eylul University. The effect of 0.75 mg (0.05 ml) and 1.5 mg (0.1 ml) of intravitreal Azyter® (THEA Laboratories, France) was compared with the intravitreal saline. Six rabbits were used for each concentration. The right eyes were designated as the study eyes. Left eyes were served as the control and same volume of saline was injected intravitreally. The rabbits were anesthetized with intramuscular ketamine hydrochloride (35 mg/kg) and xylazine hydrochloride (5 mg/kg). Proparacaine (Alcaine®, Alcon Labs, Fort Worth, TX, USA) and 5% povidone iodine were instilled. Intravitreal injection was administered with a 30-gauge needle, 1.5 mm posterior to the superotemporal limbus. A single ofloxacin eye drop 0.3% (Exocin; Allergan, Dublin, Ireland) was put on after the procedure.
At the baseline, immediately and three days after the intravitreal injection, all eyes were examined with a penlight and magnification loop and a handheld slit-lamp biomicroscope was used if needed.
Histological Analysis
The animals were sacrificed by an intravenous injection of a lethal overdose of ketamin at the third post injection week. Eyes were carefully enucleated and immediately fixed in a 10% neutral formalin. Tissue samples were embedded in parafin and five-micron thick serial sections were transferred onto the glass slides, stained with hematoxylene and eosin (H&E). Samples were examined under a light microscope (Olympus BH2, Japan) and images were transferred into a computer using a digital video camera (Olympus DP71, Japan).
Immunohistochemical Examination
A) Sections were incubated at 60˚C overnight then dewaxed in xylene. After rehydrating through a decreasing series of ethanol, sections were washed in distilled water and they were treated with 10mM citrate buffer (Cat No. AP-9003-125 Labvision), to unmask antigens by heat treatment. After that, they were cooled in buffer for 20 minutes. Sections were delineated using a Dako pen (Dako, Glostrup, Denmark) and incubated in a solution of 3% H2O2 to inhibit endogenous peroxidase activity. They were then incubated with normal serum blocking solution. Sections were then incubated in a humid chamber for 18 h at +4˚C with antibody Caspase-3 (Novus). Sections were then incubated with biotinylated IgG and then with streptavidin conjugated to horseradish peroxidase for each prepared according to kit instructions (invitrogen-Plus Broad Spectrum 85-9043). They were finally incubated with 3,30 diaminobenzidine hydrochloride (DAB) (1718096, Roche), and nuclei were counterstained with Mayer’s hematoxylin. Sections were dehydrated through a graded ethanol series, cleared in xylene, mounted in entellan (Merck, Germany) and were analyzed using a light microscope.
B) Apoptotic cell death was evaluated by in situ TUNEL (TdT-mediated dUTP-digoxigenin Nick End Labeling,Cat. No:11 684 817 910 Roche, Germany) [9]. Briefly, sections were deparaffinized, rehydrated in graded alcohol, and microwave-pretreated in trypsin solution (Roche, 10 109 819 001) at 37°C. After washing in phosphate-buffered saline (PBS), the specimens were incubated with fluorescein-labeled deoxy-UTP and TdT at 37°C for 60 min. Then, converter POD solution was applied to the slides. Sections were stained with DAB and counter-stained with hematoxylin and mounted with entellan. The percentage of TUNEL-positive cells was determined by counting the positive cells from 5 random fields in each group. The apoptotic index was defined as the number of apoptotic TUNEL-positive cells of tissues.
Statistical Analysis
All statistical procedures were performed by SPSS software for Windows, Version 15.0 SPSS, Chicago, IL, USA. All values were expressed as the mean ± standart deviation. The Mann–Whitney U-test was used to compare staining intensities values between groups. A value of p < 0.05 was considered statistically significant.
Results
None of the rabbits died during the study period and no meaningful anterior segment change was noted.
Histological and Histochemical Findings
Light microscopy demonstrated no pathological changes in the retina of all study and control eyes. However, nuclear DNA fragmentation was evident in the retinal layers of two study groups (33.6% with 1.5 mg and 21.4% with 0.75 mg azithromycin) with the TUNEL method. TUNEL staining ratio was found to be statistically higher only in the 1.5 mg azithromycin group when compared to the control group (p=0.01). The difference of TUNEL staining ratio between the two study groups was statistically significant (p=0.028).
| Eyes treated with 0.75 mg (0.05 mL) azithromycin (%) |
Eyes served as control with 0.05 mL saline injection (%) | p value | Eyes treated with 1.5 mg (0.1mL) azithromycin (%) | Eyes served as control with 0.1 mL saline injection (%) | p value | |
|---|---|---|---|---|---|---|
|
TUNEL Caspase-3 |
21.4±5.28 20.2±5.97 |
18.08±4.54 7.66±2.34 |
0.117 0.00 |
33.6±15.04 21.5±5.99 |
20.66±6.15 9.25±2.80 |
0.01 0.00 |
Caspase 3 staining was evaluated in photoreceptor cells, bipolar cells and cytoplasms of ganglion cells. The ratios of caspase-3 positive cells in two study groups were 21.5% and 20.2%, respectively. TUNEL and caspase-3 staining percentage of the eyes detected in the study are summarized in the (Table 1).
Caspase-3 staining ratio was found to be statistically higher in the eyes injected with either 0.75 mg or 1.5 mg azithromycin when compared to the control eyes (p=0.00, p=0.00 respectively).
But there were no statistically significant differences between the two study groups by caspase-3 staining (p=0.247). H&E, TUNEL and caspase-3 staining characteristics for an example eye treated with intravitreal 0.75 mg, 1.5 mg azithromycin and saline are shown in the (Fig. 1).
DISCUSSION
In everyday ophthalmology practice, intravitreal administration of antibiotics prepared from a commercial formulation is very rare and maybe the most well-known and successful clinical example of it is the administration of intracameral [10, 11] and intravitreal [12, 13] moxifloxacin 0.5% (Vigamox, Alcon Labs Fort Worth, Tx, USA). As oral azithromycin is a preferred alternative in the treatment of ocular toxoplasmosis [14] and Bartonella infections [15] intravitreal delivery may be a feasible option in the treatment armamentarium in some selected cases. Therefore toxicity concerns of intravitreal administration of azithromycin should be clarified at the first hand.
To the best of our knowledge, there is only one such study that was presented as a poster by Modarres et al. [16]. They evaluated the side effects of seven intravitreal doses of 100, 200, 300, 400, 500, 1000 and 2000 µg/0.1 cc of azithromycin in 14 albino rabbits, two rabbits for each dosage. After four weeks, the animals were sacrificed. In histopathology, the integrity and appearance of all retina layers were preserved in 100, 200, 300 and 400 µg doses. However, severe atrophic changes of all retinal layers together with cystoid degeneration and formation of a disorganized thin retina were observed in 2000, 1000 and one of 500 µg/0.1 ml injected eyes. On the other hand, we could not find any information on how the study drug was prepared or what was the findings reached in the immunohistochemical analysis.
In our study, two intravitreal doses of 0.75 and 1.5 mg were used and we found no changes in the histologic sections by the light microscopy. We analyzed the apoptosis in retinal cells by evaluating immunohistochemical staining for caspase-3 and in situ terminal deoxynucleotidyl transferase-mediated biotin-deoxyuridine triphosphate nick-end labeling of DNA fragments. With these two techniques we demonstrated that there was an apoptotic activity in the photoreceptor, bipolar and ganglion cells in the study eyes when compared to the control eyes at least in one test. This results demonstrated that intravitreal azithromycin may be harmful in the selected doses in our animal model. It is also possible that the excipient of the azithromycine (Miglyol) might also contribute to the apoptosis noted in the retinal cells. In light of our study, we still believe that there is still room for additional studies to support or refute our findings to clearly establish the side effects of intravitreal azithromycin on the retina.
CONFLICT OF INTEREST
The authors confirm that this article content has no conflict of interest.
ACKNOWLEDGEMENTS
Declared none.


