All published articles of this journal are available on ScienceDirect.
Safety and Efficacy of Myopic LASIK performed on Thin Corneas
Abstract
Purpose:
The aim was to report on the visual outcomes and safety of myopic LASIK performed in patients with corneas of central thickness below average(<540μm) and normal topography.
Methods:
This is a retrospective cohort study conducted at a private practice setting on Mexican Hispanic patients who underwent myopic LASIK between January 2014 and January 2015. An analysis of records of patients >18 years-old with previous normal topography, stable refraction, corrected visual acuity ≥ 20/20 (Snellen), Central Corneal Thickness (CCT) < 540μm and at least 12 months follow up after surgery was conducted. The main outcome measures were standard visual outcomes (efficacy, safety, refractive stability) and Percent Tissue Altered (PTA) analysis was conducted.
Results:
A total of 51 patients (102 eyes) were included; 56% (n=57) were female. The mean age was 26.52 ± 8.06 (range 18-55 years) with a mean follow up of 13.9 ± 1.2 months. Preoperative CCT was 515.44 ± 17.87μm (range 452- 540μm), with a mean refractive spherical equivalent (SEQ): -4.08 ± 2.17 D (range -0.75 to -9.75 D), and mean refractive cylinder: -1.44 ± 1.29 D (range 0.00 to -6.00 D). Mean predictability of postoperative SEQ was -0.20 ± 0.40 D (range -1.25 to +1.25). Postoperative SEQ was ±0.50 D in 71%, ±1.00 D in 93% of the eyes. Postoperative uncorrected distance visual acuity was ≥20/20 in 78% and ≥20/25 in 95%. One line of CDVA was lost in 3% of the eyes, no eyes lost ≥2 lines. No ectasia cases were observed during follow-up.
Conclusion:
LASIK surgery in Mexican Hispanic patients with thinner than “normal” corneas (<540 μm) is safe, efficient and predictable at 1 year follow up for myopic refractive corrections with no evidence of postoperative keratectasia.
1. INTRODUCTION
Laser in situ keratomileuses (LASIK) has been the treatment of choice for correcting corneal refractive errors since its introduction in early 1990 [1, 2]. Resulting in immediate high-quality visual outcomes and having excellent efficacy, predictability, stability and safety profiles, it is no wonder why LASIK surgery has become one of the today's most popular elective procedures, with more than 28 million procedures performed worldwide [3, 4]. As with any other surgical procedures, an increased frequency and widespread use are also associated with a rising incidence of complications.
Although effective methods to treat most of the complications related to LASIK have emerged (either with eye drops or with surgical correction) [5, 6] post-LASIK ectasia is one of the most feared complications since its treatment often involves extensive management strategies that range from intrastromal corneal rings [7] and crosslinking [8] to keratoplasty [9].
Specific risk factors for developing corneal ectasia after LASIK have been identified and these include deep ablation, residual stromal bed thickness lower than 300 μm, abnormal topography and Central Corneal Thickness (CCT) less than 500 μm [10-12]. Randleman et al., also considered factors as a young age and high refractive correction to develop an Ectasia Risk Score Systems (ERSS) with the objective to assess the preoperative risk for developing ectasia after LASIK [13]. Recently, the role of the Percent Tissue Altered (PTA) has been emphasized by Santhiago et al., as a robust risk indicator for developing ectasia after LASIK in the eyes with normal topography [14].
Either directly (ERSS) or indirectly (PTA), thin corneas have been considered as corneas with biomechanical liability and, therefore likely to have an increased risk for developing ectasia after ablative surgery [13, 14].
However, recent evidence shows that not only thin corneas (<500 μm) do not have an increased risk for ectasia, but that LASIK is as effective, safe and stable as in corneas with 500 μm or greater [15, 16]. In this study, we assessed the visual outcomes and safety of myopic LASIK performed in patients with corneas with central thickness below average <540 μm and normal topography.
2. METHODS
A retrospective analysis was performed on the records of Hispanic patients who underwent myopic LASIK between January 2014 and January 2015, at the Zambrano-Hellion Medical Center, Tec de Monterrey (Monterrey, México). The analysis followed the tenets of the Declaration of Helsinki; informed consent was obtained from all patients after details of the surgical procedure were explained. Inclusion criteria for the initial treatment were: age over 18 years; stable refraction with spherical component up to -8.50D, a cylindrical component up to -6.50D; corrected visual acuity ≥20/20 (Snellen visual acuity chart), a central corneal thickness (CCT) <540 μm and at least 12 months follow up.
We defined a cornea thinner than “normal” as corneas < 540 μm accordingly to the reported values (statistical mean and mode in our population. Patients with general contraindications for LASIK surgery as autoimmune disease, diabetes, pregnancy and ocular comorbidities such as glaucoma, cataract, retinal disease and dry eye syndrome were excluded. No other factors for post-refractive surgery ectasia were present.
The preoperative examination included Uncorrected Distance Visual Acuity (UDVA), Corrected Distance Visual Acuity (CDVA), manifest refraction, cycloplegic refraction, intraocular pressure measurement (Goldmann applanation tonometer), ultrasonic pachymetry (Accutome AccuPach V, Malvern, PA, USA), corneal topography (Orbscan IIz, Bausch and Lomb, Rochester, NY,USA) and slit lamp examination. The CCT was obtained using the ultrasonic pachymetry Accutome 4sight pachymeter module (Accutome, Inc., Malvern, PA,USA). Briefly, the cornea was anesthetized with topical 1% tetracaine and the patient was asked to adopt a face up position on the examination chair and solicited to fixate a target on the ceiling. The pachymeter probe was brought in contact with the cornea centrally and perpendicularly over the visual axis. CCT was recorded as the average of 9 consecutive acquisitions. This process was repeated for every individual CCT measurement.
The postoperative protocol consisted of moxifloxacin 0.5% ophthalmic solution (Vigamoxi ®, Alcon Laboratories, Fort Worth, TX, US) every 6 hours for 7 days and fluorometholone 0.1 opthalmic suspension (Flumetol, Sophia®, Jalisco, Mexico) in dose reduction for 2 weeks. Postoperative visits included UDVA, CDVA, manifest refraction, corneal topography, Goldmann tonometry, slit lamp biomicroscopy and Visante AS-OCT (Carl Zeiss Meditec Inc, Version 3.0, Dublin, CA, US) on postoperative week 1 to measure the thickness of the corneal flap.
LASIK procedures were performed by the same surgeon using a Technolas-217 Excimer workstation (Technolas Perfect Vision GmbH, München, Germany) using the standard technique. Briefly, under topical anesthesia with tetracaine chlorhydrate 0.5% (Ponti ofteno, Sophia®, Jalisco, México), the cornea was marked with gentian violet and a superior hinge was performed using a Hansatome XP Microkeratome (Bausch & Lomb, Rochester, NY). When indicated, both the eyes were operated the same day, with the refractive target to emmetropia. A 6.0 mm optical zone and a 120 microns flap with a superior hinge and an average diameter of 9.5 mm (an 8.5 mm diameter ring was used in eyes with mean keratometry > 45D) were used in every case. Zyoptix Tissue Saving-2 ablation profile was used to ensure a residual stromal bed ≥300 μm. Standard visual outcomes and Percent Tissue Altered (PTA) analysis were obtained. The preoperative and postoperative data were compared using Student's t test. Statistical analysis was implemented with the SPSS software (version 20.0, IBM Inc., NY, USA) for Windows, a p-value <0.05 was considered statistically significant. Visual acuity was measured using Snellen ́s visual acuity chart and then converted into LogMAR for statistical analysis.
3. RESULTS
A total of 51 patients (102 eyes) were included in the study, 56% (n=57) were female. The mean age was 26.52 ± 8.06 (range 18 to 55 years) with a mean follow up of 15.9 ± 1.2 months. Preoperatively, CCT was 515.44±17.87μm (range 452-539μm), the mean refractive spherical equivalent (SEQ) was -4.06±1.85D (range -0.75 to -9.75D) with a mean refractive cylinder of -1.44 ± 1.29D (range 0.00 to -5-75D). On postoperative week 1, the mean central thickness of the corneal flap was (128.66 ± 17.09 μm). The analysis of PTA showed a mean value of 0.35 ± 0.04 (range 0.22 to 0.44). Fig. (1) shows the Standard Graphs for Reporting Refractive Surgery.
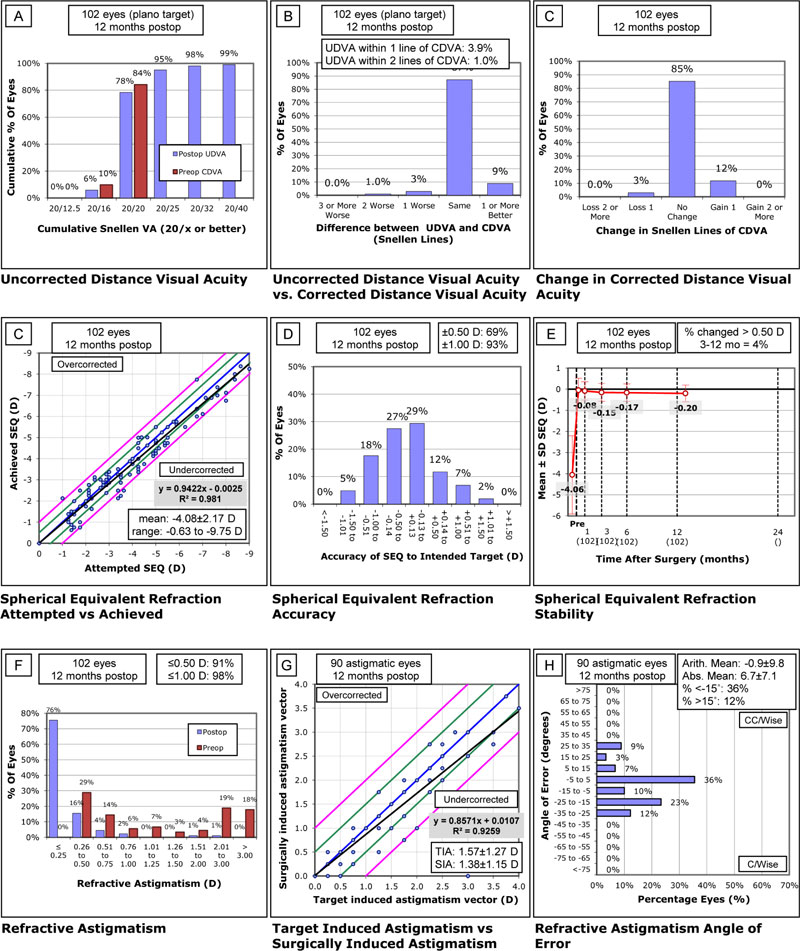
The mean predictability of postoperative SEQ was -0.20 ± 0.40 D (range -1.25 to +1.25) at the end of the follow-up. Postoperative SEQ was ±0.50 D in 71% and ±1.00 D in 93% of the eyes. Preoperative CDVA was 20/20 or better in 93% of the eyes. Postoperative uncorrected distance visual acuity was 20/20 or better in 78% and 20/25 or better in 95% of the eyes. One line of CDVA was lost in 3% of the eyes and none of the eyes lost more than 2 lines of CDVA. Over the follow up (from postoperative month 3 to postoperative month 12), only 4% of the eyes changed >0.50D. A strong squared correlation (R2=0.981) was observed between attempted and achieved SEQ correction. (Table 1) shows the changes in visual and refractive outcomes before and after the LASIK procedure. Intraoperative complications consisted of epithelial defect in 3 cases (3% of total) and flap striae that required flap re-lifting in 1 eye (1%). No ectasia cases were observed during follow-up.
| Visual and Refractive Outcomes Before and After Myopic LASIK in Thin Corneas | ||||
|---|---|---|---|---|
| Parameter | UDVA (LogMAR) a | CDVA (LogMAR) a | SEQ (D) a | Keratometry (D) a |
| Preoperative | 0.84 ± 0.45 | 0.00 ± .05 | -4.06 ± 1.85 | -1.44 ± 1.29 |
| 6 months FwUp | 0.00 ± .08 | 0.00 ± .04 | -0.17 ± 0.41 | -0.47 ± 0.40 |
| End point FwUp | 0.00 ± .05 | 0.00 ± .02 | -0.20± 0.40 | -0.36 ± 0.39 |
| P valueb | <.001 | <.001 | <.001 | <.001 |
| P valuec | .78 | .81 | .13 | .08 |
4. DISCUSSION
Post LASIK ectasia is rare, but even with a prevalence rate of 0.02% to 0.6%, it remains as one of the most feared complications in refractive surgery [17, 18]. Risk factors for developing this condition have been previously identified [12, 14], amongst them, thin corneas (<500 μm) have been historically considered as corneas with biomechanical frailty and therefore as corneas predisposed to develop ectasia [19, 20]. Evidence shows that factors as race [21, 22], age and gender [22, 23] altitude [24] and UV light exposure [25] may influence CCT, hence different “normal” corneal thicknesses have been established amongst various research groups. In a meta-analysis conducted by Doughty et al. [23], an average CCT of 536±29 μm was established for normal healthy eyes. In a Hispanic population, our group observed a mean CCT of 545.69 ± 36.88 μm (mode of 540 μm) in healthy corneas of Hispanic patients [26]. In this study, we evaluated the visual outcome, safety and predictability of LASIK performed on a large cohort of corneas thinner than “normal”, defining the latter as corneas <540 μm accordingly to the reported values (statistical mean and mode) in our population, at a 12 month follow up.
Against the old paradigm that thin corneas have a biomechanical liability, recent evidence has shown not only the absence of keratectasia during follow up but also no difference in visual outcomes, safety and predictability when LASIK is performed on thin corneas with normal topography when compared with preoperative corneas with average or normal thickness. Tomita et al., assessed the 6 year-follow up outcomes of thin-flap LASIK in eyes with thin corneas (CCT<500 μm) but normal topography and compared them with the outcomes of LASIK performed on corneas with CCT 500 μm or greater [16]. They observed no difference in visual, refractive and topographic outcomes at the long-term between both groups. At their last follow-up, 83% of the eyes in the thin cornea group achieved a UDVA of 20/20 or better, 63% were stable or gained lines of CDVA and had refractive stability with a MRSE change of -0.17 ± 0.42 D over time [16]. Similarly, we observed 78% of the patients with UDVA ≥20/20, 97% of the eyes were stable or gained lines of CDVA at the last follow up and refractive stability with a MRSE change of -0.20 ± 0.40 over time. Likewise, we observed a non-significant difference in visual and refractive outcomes when comparing 6-month follow-up with the final follow up (Table 1), suggesting visual and refractive stability.
Caster et al., performed a retrospective analysis of 109 eyes with a preoperative central corneal thickness of ≤500 μm and otherwise normal topography that underwent LASIK, having a postoperative follow up of at least 12 months [15]. As in the study by Tomita et al. [16], and the present study, refractive stability was observed during the follow-up period with no incidence of postoperative keratectasia. Previously, Binder et al., examined a database of 9700 eyes that underwent myopic LASIK and he found 117 eyes with corneal pachymetry <500 microns and a follow up of at least 2 years with no report of corneal ectasia [17]. Kymionis et al., also showed the results of 124 eyes with thin corneas less than 500 microns that underwent excimer laser cornea refractive surgery (either PRK or LASIK) observing good predictability (mean predictability of 0.08 ± 0.40 D for PRK group, 0.14 ± 0.55D for the LASIK group) and no ectasia during the follow-up (1 year).
Corneal thickness has been considered as an inherent sign of structural stability; hence different authors have included thin corneas as a risk factor to develop postoperative keratectasia after excimer laser corneal refractive surgery [13, 17-29]. However, the question if thin corneas should be considered as “weak” corneas and there as an independent risk for post-LASIK ectasia is yet in dispute. Recent evidence, including the present study, has failed to categorize thinner than normal corneas as an independent risk for developing keratectasia after LASIK or PRK, since not only thin corneas perform as efficiently and safely than normal thickness corneas after refractive surgery but they did not show a trend over time to evolve in to ectasia. Focusing on a flap thickness tailored to the initial corneal thickness and to the amount of ablation has been a more important issue on the debate, since the evidence from the work of Santhiago et al. [14], has shown that the percent of tissue altered ≥40% (obtained from the quotient of the sum of flap thickness and ablation depth over the central corneal thickness) was a more robust indicator that other individual variables (included CCT <510 μm) for the development of corneal ectasia after LASIK in eyes with normal topography.
In our series, a mean PTA of 0.35 ± 0.04 was achieved and although the recommendation in these patients is to create flaps of precise thickness using the femtosecond laser, we observed an acceptable flap thickness using a mechanical microkeratome (postoperative flap thickness 128.66 ± 17.09 μm).
A weakness of this study is its retrospective nature and the limited follow up (mean follow up of 15.9 ± 1.2 months). However it is a large retrospective cohort of patients’ eyes with thinner than “normal” corneas and normal topography that underwent LASIK and along with previous studies of Caster [15] (109 eyes), Kymionis [27] (56 eyes with LASIK and 68 with PRK), Binder [11] (107 eyes) and Tomita [16] (291 eyes, case control), it contributes with evidence arguing against thin corneas as an independent risk factor for keratectasia after ablative corneal surgery. A control group with normal corneas for our population (≥540 μm) could also potentially enhance the power of the study by eliminating or isolating confounding variables and bias.
CONCLUSION
In conclusion, we observed that LASIK surgery in patients with corneas thinner than “normal” (<540 μm) is safe, efficient and predictable at 1 year follow up for myopic refractive corrections with no evidence of postoperative keratectasia. Evidence in this and similar works suggest that LASIK surgery in eyes with preoperative thinner than normal cornea and normal topography may not be a risk factor if a fair residual stromal bed (at least 300 μm) and a PTA <40% is ensured. Longer follow up and a larger cohort of patients are needed to support and reinforce the proposition that thinner than normal corneas perform as efficiently and safely than normal thickness corneas after excimer refractive surgery.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
This study was approved by the Tecnologico de Monterrey School of Medicine (Monterrey, Mexico) Ethics and Research Committee under approval #IRB00011357.
HUMAN AND ANIMAL RIGHTS
No animals were used in this study. All human research procedures followed were in accordance with the Helsinki Declaration of 1975, as revised in 2013.
CONSENT FOR PUBLICATION
Informed consent was obtained from all the participating patients.
AVAILABILITY OF DATA AND MATERIALS
The datasets used and/or analyzed during the current study can be made available on reasonable request to the corresponding author [J.G].
FUNDING
None.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Declared none.


