All published articles of this journal are available on ScienceDirect.
Clinical Outcomes of Laser Asymmetric Keratectomy to Manage Postoperative Adverse Effects–A Retrospective Clinical Trial
Abstract
Background:
Laser asymmetric keratectomy reduces the regional asymmetry of corneal thickness.
Objective:
We aimed to describe the clinical outcomes of laser asymmetric keratectomy keratectomy with laser refractive surgery performed to resolve the adverse effects following ophthalmic surgeries.
Methods:
We compared the preoperative and postoperative outcomes and complaints of blurring after performing laser asymmetric keratectomy with laser refractive surgery in 24 eyes of 16 patients with a deviation sum in corneal thickness in four directions >80 µm. Laser asymmetric keratectomy with laser refractive surgery, with full integration of the Vision Up software, was used to analyze the corneal thickness deviation, employed selective laser ablation to create central symmetry on the thicker cornea to reduce regional asymmetry of corneal thickness, simultaneously correcting the refractive power and myopic shift. The pre-and postoperative clinical and topographic findings were analyzed.
Results:
The patients’ age was 37.57±22.30 (range, 23–65) years. The follow-up period was 16.56±3.23 months. The spherical equivalent (p=0.026), sphere (p=0.022), uncorrected distance visual acuity (LogMAR, p=0.045), blurring score (p=0.000), central corneal thickness (p=0.024), sum of deviations in corneal thickness in four directions (p=0.02), distance between the maximum posterior elevation and visual axis (p=0.04), and kappa angle (p=0.031), significantly decreased postoperatively. The efficiency and safety indices were 0.96±0.11 and 1.00, respectively. There was no myopic regression or blurred vision postoperatively.
Conclusion:
Performing laser asymmetric keratectomy with laser refractive surgery improved corneal symmetry and visual acuity and reduced blurring.
1. INTRODUCTION
After undergoing ophthalmic surgeries, patients usually complain of blurred vision, reduced visual acuity, discomfort while driving at night, worsening dryness of the eye, and blurring of vision [1-9]. Although studies have examined ways to reduce the adverse effects of laser refractive surgery (LRS), the results have shown little differences compared with regular LRS [6, 7, 10, 11]. Conventional LRS symmetrically ablates the cornea. In cases of regional asymmetry of the corneal thickness, the postoperative thinner portion of the cornea can be protruded by the intraocular pressure (IOP); therefore, new customization methods are needed [12-14]. Previous studies have reported that these new techniques can prevent postoperative corneal distortion [15-20]. Adverse effects, such as blurring or myopic regression after various ophthalmic surgeries, may occur because of corneal asymmetry of corneal thickness [21-26]. Laser asymmetric keratectomy (LAK) has been reported as a type of customization that achieves corneal symmetry and satisfactory surgical results [21-26]. The present study aimed to report the surgical outcomes of LAK to resolve complications, such as reduced visual acuity and blurring, which occur because of corneal asymmetry (SUM >80 µm) after undergoing various types of ophthalmic surgery.
2. MATERIALS AND METHODS
This retrospective study reviewed the pre-and postoperative outcomes of LAK-LRS (LASIK, LASEK) [21-24], performed by the same surgeon at the Woori Eye Clinic in Daejeon, the Republic of Korea, between January 2018 and August 2019. Especially, we examined the data of patients who were available for follow-up examinations at 1 year postoperatively.
The inclusion criteria of this retrospective study were as follows: 1) performance of LAK-LRS between January 2018 and August 2019; 2) SUM ≥80 mm; 3) Patients’ complaints of blurring and or myopic regression after undergoing ophthalmic surgeries; and 4) ≥1 year follow-up period. The exclusion criteria were 1) history of corneal disease and trauma, and 2) follow-up period of <1 year.
| Items | Findings |
| Preoperative | |
| Age (years) | 37.57±22.30 (range: 23-65) |
| Male: Female ratio | 8:8 |
| Previous operations: patients (eyes) LASIK LASEK Multifocal IOL (Cataract op) Trabeculectomy Interval (months) |
6 (8) 6 (11) 2 (3) 2 (2) 36.03±20.70 (range:16-68) |
| Intraoperative findings | |
| Optic zone (mm) | 6.33±1.70 |
| Ablation depth (µm) | 57.62±16.61 |
| Myopic shift due to LAK | -1.93±1.54 |
| Residual stromal depth (µm) | 399.73±20.40 |
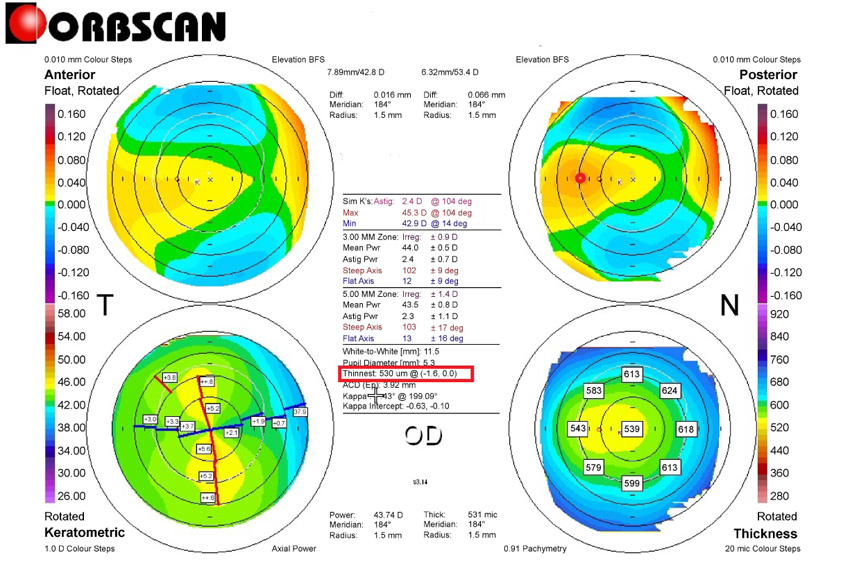
An example of the right lower map measuring the differences in thickness between the symmetrically opposed points at eight locations at 2.5 mm from the center of the cornea (i.e., at 0º, 45º, 90º, 135º, 180º, 225º, 270º, and 315º); then, the SUM value was calculated (0–180º, 45–225º, 90–270º, and 135–315º). In the presented case, the results were as follows: 0–180º, 75 µm; 45–225º, 45 µm; 90–270º, 14 µm; and 135–315º, 30 µm; total SUM, 164 µm. Right upper map: measuring DISTANCE. The corneal apex was temporally deviated (red circle). The thinnest point (X, Y) is indicated with the lower red square. DISTANCE, distance between the maximum posterior elevation (best-fit-sphere [BFS]) and the visual axis; SUM, sum of deviations in corneal thickness in four directions on the Orbscan map.
In total, 16 healthy patients (24 eyes) complained of blurred vision following ophthalmic surgery. Six (8 eyes), 6 (11 eyes), 2 (3 eyes), and 2 (2 eyes) patients complained of blurring after LASIK, blurring after LASEK, blurring after cataract surgery and multifocal intraocular lens insertion, and blurring after glaucoma and trabeculectomy surgery, respectively (Table 1).
Furthermore, in terms of blurring, 8, 5, and 3 patients experienced halos, glares, and starbursts, respectively. This study was conducted in accordance with the 1964 Declaration of Helsinki and its later amendments and was approved by the Korean National Institute for Bioethics Policy (approval number: P01-201911-21-011). Written informed consent to publish clinical patient information and photographs was obtained from the patients
Patients with refractive error underwent LAK-LRS (LASIK or LASEK) using a 193-nm ISOD200 laser device (Kera Harvest Inc., Taibao City, Taiwan). Vision-Up software (WellC, Seoul, Republic of Korea) [20, 23, 24] was used to analyze the central corneal thickness (CCT) deviation based on the Orbscan II (B&L) corneal maps (Fig. 1). The software is also used to inform the laser concerning the thicker corneal regions that should be ablated to ensure corneal symmetry, as determined by LAK. In addition, Vision-Up software predicts myopic shift because of LAK (Fig. 2). Therefore, the surgeon could create a laser ablation plan to offset the corneal change when the laser eliminates the center thickness deviation by blocking the thinner corneal regions. Overall, Vision-up analysis allows the achievement of central symmetry and satisfactory surgical outcomes of the refractive power (Fig. 3) [21, 23-26].
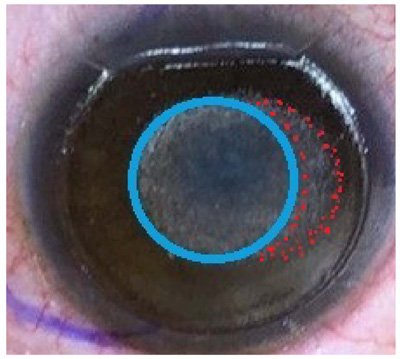
A LAK ablation pattern (total of -3.75 diopters) for the refractive error (red dots, -1.75 diopters) and myopic shift (blue circle, -2.0 diopters). The area enclosed in red dots indicates an asymmetric LAK ablation pattern on a thicker area of the cornea in the peripheral region.
LAK, laser asymmetric keratectomy.
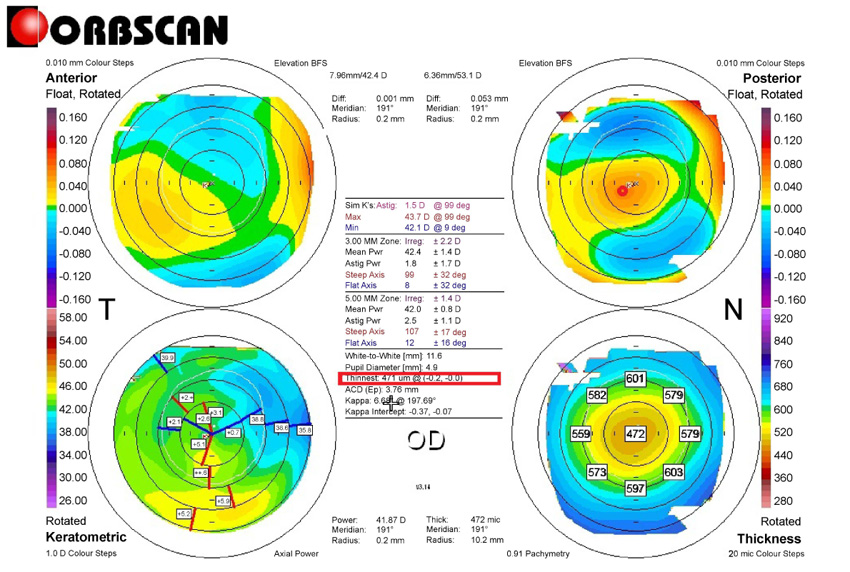
Right upper and lower maps following the same representation conventions as in Fig. 1. In the presented case, the results were as follows: 0–180º, 20 µm; 45–225º, 6 µm; 90–270º, 4 µm; and 135–315º, 21 µm; total (SUM), 51 µm. DISTANCE was measured as in Fig. 1.
DISTANCE, distance between the maximum posterior elevation (best-fit-sphere [BFS]) and the visual axis; SUM, sum of deviations in corneal thickness in four directions on the Orbscan map.
LAK-LRS improved corneal symmetry by reducing SUM and correcting the refractive power (myopia and astigmatism) and myopic shift simultaneously (Figs. 2 and 3).
In LAK-LRS, the optic zones, central corneal ablation depths, myopic shifts, and residual stromal beds were checked intraoperatively.
During the pre- and postoperative examinations, the extent of blurring with the correction of refractive errors was classified as a score ranging from 0 to 3 points (0, none; 1, mild; 2, moderate; 3, severe or disturbing) [4] based on a medical interview performed before and after LAK performance (Table 2).
Pre- and postoperative uncorrected distance visual acuity (UDVA), spherical equivalents (SEs), sphere, cylinder, blurring scores, IOP, CCT, SUM, visual axis (DISTANCE), kappa angle, corneal irregularity at the 3.0-mm and 5.0-mm zones on the Orbscan map were analyzed retrospectively. The refractive power was measured using an auto-refract-keratometer (and calculated as the SE). Myopic shift during the follow-up period was defined as a change in myopia indicated by an SE ≥1.0 diopter after surgery. The SUM and DISTANCE values on the Orbscan map were measured using the same methods [21, 23, 24].
For each eye, we calculated an efficiency index (postoperative uncorrected visual acuity/preoperative best spectacle-corrected visual acuity [BSCVA]), safety index (postoperative BSCVA/preoperative BSCVA), and percent postoperative myopic regression.
2.1. Statistical Analysis
The Statistical Package for the Social Sciences (SPSS Inc., Chicago, IL, USA) was used for statistical analyses. Paired t-tests were performed, and p-values <0.05 were considered statistically significant. Data are presented as means±standard deviations unless otherwise noted.
3. RESULTS
A total of 16 otherwise healthy patients (8 male and 8 female patients; 24 eyes) complained of blurring or myopic regression following ophthalmic surgeries. The mean age of the patients was 37.57±22.30 (range, 23–65) years. The time interval from the performance of ophthalmic surgery (LASIK, LASEK, cataract operation, trabeculectomy) to that of LAK-LRS was 36.03±20.70 (range, 16–68) months.
The intraoperatively optic zone was 6.33±1.70 mm, the ablation depth was 57.62±16.61 µm, the myopic shift because of LAK was -1.93±1.54 D, and the residual stromal depth was 399.73±20.40 µm (Table 1).
| Score | Extent of Blurring |
| 0 | No blurring |
| 1 | Mild blurring, able to perform daily activities and drive at night |
| 2 | Moderate blurring, discomfort in daily activities and while driving at night |
| 3 | Severe blurring, severe discomfort in daily activities and unable to drive at night |
In the comparison between the preoperative and postoperative findings of LAK (Table 3, Figs. 1 and 3), the UDVA (Log MAR) was 0.11 ± 0.19 (over 20/25 in Snellen acuity), respectively. Postoperatively, the following variables markedly decreased: the SE (p=0.026), sphere (p=0.022), UDVA (LogMAR; p=0.045), SUM (p=0.02), DISTANCE (p=0.04), kappa angle (p=0.031), CCT (p=0.024), blurring score (p=0.000). No significant change was observed in the following variables: cylinder (p=0.348), IOP (p=0.567), corneal irregularities in 3.0-mm (p=0.364) and 5.0-mm zones (p=0.360) on the Orbscan map, and pupil size (p=0.472).
The postoperative myopic regression rate was 0% and the efficiency index was 0.96 ± 0.11. The safety index was 1.00 (Table 3). Postoperatively, there were no significant blurrings, such as halos or starbursts.
4. DISCUSSION
In the present study, Vision-Up software increased the surgical success rate [21, 23-26]. The software was very useful for analyzing the corneal thickness deviations observed on the Orbscan II corneal maps. Especially, it predicted the target amounts of ablation at thicker regions of the cornea, the direction in degrees, and the amount of the expected corneal myopic change after performing the LAK procedure. The high precision of the output led to satisfactory surgical results with LAK-LRS.
All 12 patients (19 eyes) who complained of blurring after LASIK or LASEK showed SUM values of ≥80 microns; therefore, we believe that the thinner parts of the cornea showed a higher refractive power compared to other areas, leading to a more negative posterior curvature value, thus, resulting in myopic regression. The discrepancy in curvature between the anterior and posterior surface results in a difference in refractive power, which causes light entering through the pupil to scatter, thus, resulting in blurring. After performing LAK, the SUM, DISTANCE, and blurring scores markedly decreased as the cornea became more symmetrical postoperatively. In general, phacoemulsification surgeries do not change the corneal shape. Nevertheless, some types of surgery could cause blurring symptoms because of increased corneal asymmetry postoperatively. Preoperative lens control completely accommodated, but disappears after lens removal by cataract surgery and vision may become blurred by the asymmetric cornea.
The 3 eyes that developed blurring after undergoing cataract surgery with multifocal intraocular lens insertion also showed similar findings to those observed in the patients who underwent LASIK or LASEK concerning SUM and DISTANCE. After performing cataract surgery, the decrease in the IOP was approximately 2 mmHg, and factors, such as reduced corneal stiffness, were reported to be a cause of biomechanical changes [8, 27, 28]. After performing LAK, the blurring and the visual acuity improved, also showing values of SUM <80 µm on the Orbscan map, which is encouraging for the treatment of visual abnormalities caused by corneal deformation after cataract surgery. The two eyes with blurring after trabeculectomy for glaucoma showed very large SUM values. An aqueous channel is made in the sclera, and the scleral filtering flap causes traction on the cornea; the postoperative decrease in IOP probably led to corneal deformation and changes in corneal thickness which in turn caused severe blurring and visual impairment. After performing LAK, the SUM, DISTANCE, and blurring scores markedly decreased. Therefore, in the event of blurring and visual impairment after glaucoma surgery, it is important to perform a topographic examination to determine whether there is any corneal deformation [9]. Although we did not measure the optical aberration with an oberrometer, we observed decreased SUM, DISTANCE, and kappa angle values and no significant quantitative increases in corneal irregularities in the 3.0-mm zone on the Orbscan maps postoperatively. These outcomes may be attributed to the excellent corneal point symmetry effect of the LAK technique.
We employed the Orbscan maps for corneal topography visualization rather than the current Sheimpflug techniques. DISTANCE can be calculated only on an Orbscan map because these maps feature a central visual axis and, thus, they are very useful for measuring symmetry.
The performance of LAK has been reported recently [21, 23-26]. Moreover, unlike previously reported techniques, such as wavefront- and topography-guided LASIK or LASEK [6, 7, 10, 11], it reduces corneal thickness deviations by asymmetric corneal ablation. Previous studies have reported that the performance of LAK has several advantages. In case of the asymmetric cornea after wavefront- and topography-guided LASIK or LASEK, the shape of the cornea can be changed to reduce the refractive errors [6, 7, 10, 11]. However, as LAK is an asymmetric ablation of the cornea at the center of the cornea to eliminate corneal thickness deviation, it has two advantages, corneal symmetry can be improved postoperatively, and refractive power is also corrected [21, 23, 24].
Further, it increases the corneal symmetry by decreasing the SUM and DISTANCE metrics. Therefore, postoperative prevention of corneal changes is expected [15-26].
Since LAK has reported been recently [21, 23-26], future studies on LAK and its treatment outcomes are needed. LAK, which was developed based on the concept that symmetrical morphology results in fewer optical aberrations than asymmetrical morphology, aims to create symmetrical corneal thickness around the center of the cornea and has been shown to improve optical aberrations [21-26].
CONCLUSION
In patients complaining of blurred vision following ophthalmic surgery, the performance of LAK could reduce the SUM and DISTANCE values, thereby increasing the central symmetry of the cornea, ultimately resulting in reduced blurring.
LIST OF ABBREVIATIONS
| LAK-LRS | = Laser Asymmetric Keratectomy with Laser Refractive Surgery |
| SUM | = Sum of Deviations in Corneal Thickness in four Directions on the Orbscan Map |
| SE | = Spherical Equivalent |
| UDVA | = Uncorrected Distance Visual Acuity |
| LogMAR | = Logarithm of the Minimum Angle of Resolution |
| CCT | = Central Corneal Thickness |
| DISTANCE | = Distance between the Maximum Posterior Elevation (best-fit-sphere) and the Visual Axis |
| IOP | = Intraocular Pressure |
| LAK | = Laser Asymmetric Keratectomy |
| LRS | = Laser Refractive Surgery |
| LASEK | = Laser Epithelial Keratomileusis |
| LASIK | = Laser in situ Keratomileusis. |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
This study was approved by the Korean National Institute for Bioethics Policy (approval number: P01-201911-21-011).
HUMAN AND ANIMAL RIGHTS
No animals were used in this research. All human research procedures followed were in accordance with the ethical standards of the committee responsible for human experimentation (institutional and national), and with the Helsinki Declaration of 1975, as revised in 2013.
CONSENT FOR PUBLICATION
Written informed consent to publish clinical patient information and the photograph was obtained from the patients.
AVAILABILITY OF DATA AND MATERIAL
The datasets generated and/or analyzed during the current study are not publicly available but are available from the corresponding author on reasonable request.
FUNDING
None.
CONFLICT OF INTEREST
The author declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
We would like to thank Editage (www.editage.co.kr) for English language editing.


