All published articles of this journal are available on ScienceDirect.
Azzopardi Phenomenon in a Non-neoplastic Phthisical Eye: A Case Report
Abstract
Objective:
The Azzopardi phenomenon, known as the deoxyribonucleic acid deposition on various structures due to cellular necrosis, has never been reported in non-neoplastic eyes.
Methods:
We report a case of a 48-year-old man who had congenital nystagmus with poor vision in both eyes, presented with decreased vision and photophobia in his left eye. An exudative retinal detachment was found, which did not respond to systemic steroid treatment. Glaucoma due to occlusio pupillae was later developed. Laser iridotomy and anti-glaucoma medications decreased intraocular pressure to an acceptable level. Vision in the left eye gradually deteriorated during the 10-year clinical course. Evisceration was finally performed due to persistent dull aching ocular pain along with signs of ocular hypotony
Results:
Histopathological examination showed phthisis bulbi and focal nodular retinal gliosis. The Azzopardi phenomenon was found at the retinal vessel walls, within the retinal layers and along the internal limiting membrane. There was neither evidence of intraocular tumors nor foreign bodies.
Conclusion:
This case demonstrated that the Azzopardi phenomenon could be present in a non-neoplastic eye with a longstanding disease that proceeds to phthisis bulbi.
1. INTRODUCTION
The Azzopardi phenomenon, or the Azzopardi effect, was first described by John G. Azzopardi in 1959 [1]. The hematoxyphil (basophilic stained) sheaths around blood vessel walls at the necrotic foci of the oat-cell carcinoma, which was initially believed to be calcification, were first proven by Azzopardi to be the deposition of breakdown products of deoxyribonucleic acid (DNA) liberated from degenerated tumor cells. The material displayed a positive Feulgen reaction and was negative with stains for calcium. He found that almost one-third of oat-cell (small cell) carcinoma specimens had demonstrated this finding.
Most of the reported cases with the Azzopardi phenomenon were from rapidly growing and degenerating tumors, such as pulmonary small cell carcinoma [1, 2], extra-pulmonary small cell carcinoma [3-5], Burkitt’s lymphoma [6], medulloblastoma [7], ependymoblastoma [7], Merkel cell carcinoma [8, 9], primary high-grade neuroendocrine carcinoma of the esophagus [10], squamous cell carcinoma, and polymorphous low grade adenocarcinoma of the oral cavity and the salivary glands [11].
The Azzopardi phenomenon was encountered in a few non-neoplastic cases, i.e., in metal-on-metal arthroplasty cases unrelated to malignancy [12]. This report was later questioned as it was thought to be iron deposition rather than DNA deposition [13]. The phenomenon is also found in functioning parathyroid microcysts, reported in two patients presenting with hyperparathyroidism and hypercalcemia [14]. The proposed mechanisms of these findings were rapid cell cycling or extensive cytoplasmic shrinkage.
Regarding the ophthalmic pathology, Mullaney, in 1968, had originally described this phenomenon in retinoblastoma eyes [15, 16] and later elaborated that the DNA deposition around blood vessels could be found in necrotic areas, calcified foci, cellular areas, and anterior segment of retinoblastoma specimens [17]. Stowe et al. in 1979 had studied the incidence of the Azzopardi phenomenon (so-called vascular basophilia) in ocular and orbital tumors. They observed this phenomenon in 6% of retinoblastoma and 1.2% of metastatic carcinoma to the eye and orbit. Oat-cell carcinoma was found to be the major primary cause [18].
As far as we are aware, there have been no previous observations of the Azzopardi phenomenon in a non-neoplastic eye. We herein report this unusual phenomenon in a phthisical eye without evidence of intraocular tumor nor intraocular metal foreign body.
2. CASE PRESENTATION
A 48-year-old male patient, who had a history of congenital nystagmus and poor vision, partially improved with corrective spectacles, was presented to the eye clinic with decreased vision and photophobia in his left eye. He had neither history of ocular trauma nor intraocular surgery. His corrected visual acuity with spectacles (-10.0 D for the right eye and -10.75 D for the left eye) was 20/200 in the right and 5/400 in the left. Intraocular pressure was 21 and 12 mmHg, respectively. Right eye examination appeared to be normal with insignificant retinal pigmentary epithelial changes at the periphery. Left eye examination showed clear cornea, a few cells with flare in the anterior chamber, and exudative retinal detachment. Oral prednisolone 60 mg per day was administered. The retinal detachment did not subside. At a one-month follow up visit, left eye intraocular pressure increased to 38 mmHg from the occlusio pupillae. Iris bombe and high pressure were treated by peripheral laser iridotomy and anti-glaucoma medications. At 6 months, the vision continued to deteriorate and total retinal detachment was observed. At 4 years, the patient had no light perception in the left eye, but the eye was not painful. At 8 years, the intraocular pressure increased again to 35 mmHg. The eye examination showed total occlusion of the pupil, iris neovascularization, localized iris bombe at 2 and 6 o’clock, and the presence of a cyclitic membrane. Cyclophotocoagulation was then planned but was not performed due to well-controlled intraocular pressure after laser iridotomy at the iris bombe areas along with increments of anti-glaucoma medications. The anti-glaucoma eye drops were finally tapered off. At 10 years, the patient reported persistent dull aching pain in the left eye even with the low intraocular pressure (range, 2 to 7 mmHg). The patient agreed to undergo evisceration to relieve the symptoms. Pre-operative physical examination showed no systemic diseases. Complete blood count and chest X-Ray were within normal limits. At a 6-month follow up examination after the operation, the patient was doing well and satisfied with the results. There was no evidence of implant extrusion or other systemic diseases.
3. RESULTS AND DISCUSSION
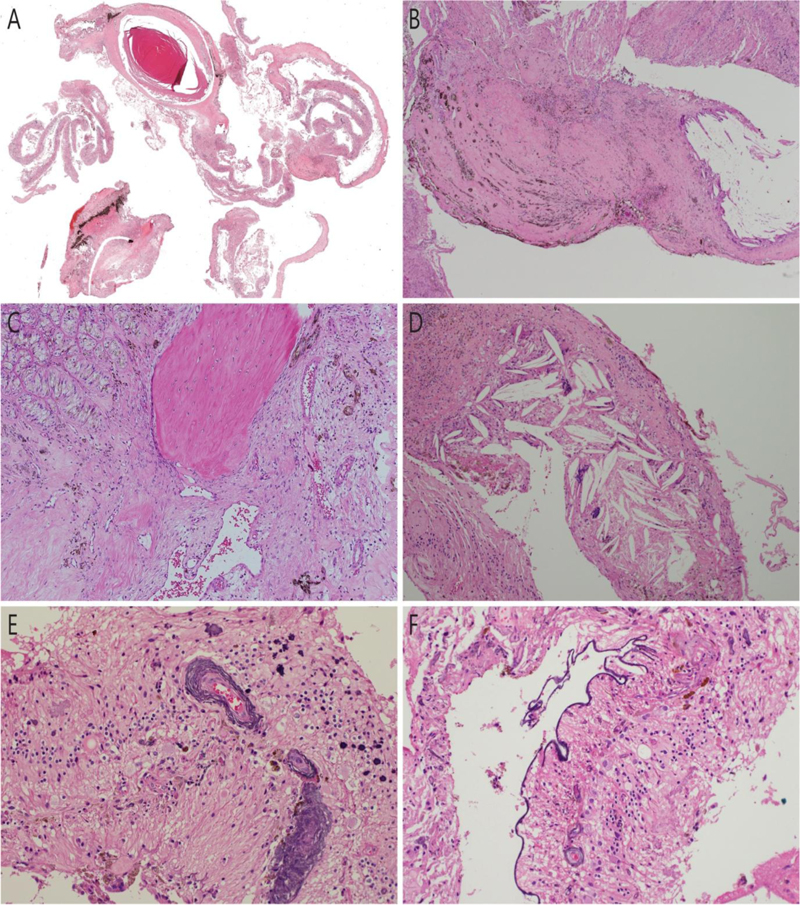
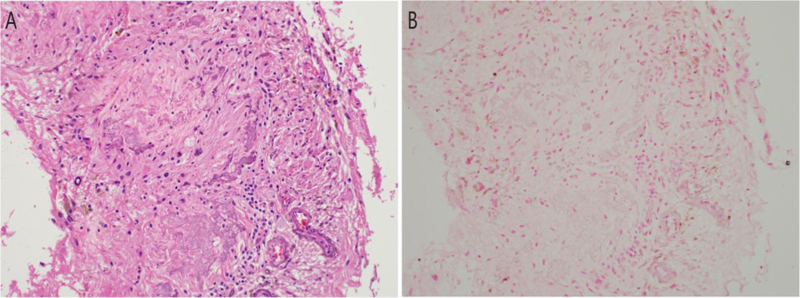
Histopathological examination of the evisceration specimen showed poorly identifiable structures of the eyeball (Fig. 1A). A neovascular membrane at the anterior surface of the iris, partially calcified lens, and a few small lymphoid aggregates in the choroid were observed. The retina was disorganized, and it showed a localized glial mass representing fibrous metaplasia, occupying approximately one-fifth of the vitreous cavity (Fig. 1B) along with small foci of osseous metaplasia (Fig. 1C) and multinucleated giant cells in the vicinity of numerous cholesterol clefts (Fig. 1D). Deep basophilic staining encircling the retinal vascular channel walls as well as scattered small basophilic clumps of varying sizes were widespread among the poorly identifiable retinal layers (Fig. 1E). Linear deep basophilic staining of the internal limiting membrane was also found (Fig. 1F). The specimen did not show any viable or necrotic tumor cells. There was no evidence of intraocular foreign bodies. The intraretinal basophilic clumps (Fig. 2A) were negative by von Kossa stain for calcium (Fig. 2B). Perl’s Prussian Blue stain for iron was negative. The final pathological diagnosis was phthisis bulbi with focal nodular retinal gliosis [19], and the Azzopardi phenomenon at the posterior part of the eyeball, i.e., around the retinal vessels, within the retinal layers, and along the internal limiting membrane.
Although the Azzopardi phenomenon is not unexpected in rapidly growing tumor, this phenomenon is extremely rare in non-neoplastic cases. Reports in non-neoplastic lesions showed this effect in only a few cases with metal-to-metal interaction and rapidly growing cells in functioning parathyroid microcysts [12-14].
We searched for the literature using the keywords “Azzopardi phenomenon” OR “Azzopardi effect” in PubMed, Scopus, and Google Scholar databases; there has been no previous report of this phenomenon in non-neoplastic eyes.
The presence of cyclitic membrane, lens calcification, focal nodular retinal gliosis, and cholesterol clefts represented a prolonged course of the disease. We did not find any evidence of tumor cells or intraocular foreign bodies in this eye.
CONCLUSION
This case report is an example of the Azzopardi phenomenon in longstanding ophthalmic pathology with evidence of inflammation and possibly from extensive cellular necrosis during the prolonged clinical course of the disease. We, therefore, conclude that the Azzopardi phenomenon can be present in non-neoplastic eye that proceeds to phthisis bulbi.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The research ethics review was exempted by the Institutional Review Board of the Faculty of Medicine, Chulalongkorn University (Certificate of exemption number 001/2021).
HUMAN AND ANIMAL RIGHTS
The report adhered to the tenets of the Declaration of Helsinki as amended in 2008.
CONSENT FOR PUBLICATION
Written informed consent was obtained from the patient.
AVAILABILITY OF DATA AND MATERIALS
The authors confirm that the data supporting the findings of this study are available within the article, and further information is available from the corresponding author, [W.T.], on special request.
STANDARDS OF REPORTING
CARE guidelines have been followed in this study.
FUNDING
None.
CONFLICTS OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
We thank Ms. Sranya Phaisawang and the English editing service, Research Affairs, Faculty of Medicine, Chulalongkorn University, for assistance in the manuscript.


