All published articles of this journal are available on ScienceDirect.
Intracameral Recombinant Tissue Plasminogen Activator for Refractory Glaucoma Secondary to a Fungal Corneal Abscess
Abstract
We present a 78-year-old patient who developed refractory glaucoma following a fungal infection of the corneal incision after cataract extraction with posterior chamber intraocular lens implantation. An anterior chamber injection of 0.1 ml recombinant tissue plasminogen activator (25 mcg/0.1 mL) was performed to improve the trabecular meshwork outflow facility compromised secondary to fibrin blockade. The intraocular pressure dropped dramatically from 48 mmHg to 10 mmHg postoperatively, negating the need for glaucoma surgery.
INTRODUCTION
Recombinant tissue plasminogen activator (rTPA) in ophthalmology has been used in the treatment of hyphema [1] and fibrin formation after cataract surgery [2, 3].In glaucoma surgery, rTPA has been used for tube shunt occlusion, bleb failure and fibrin effusion [4, 5].
We present a case of refractory glaucoma following a fungal infection of a corneal incision after cataract extraction that was successfully treated with intracameral injection of rTPA.
CASE REPORT
A 78-year-old Caucasian female presented with a red painful left eye at her 1 month postoperative visit after uneventful cataract extraction with posterior chamber intraocular lens implantation. The visual acuity (VA) was 20/20 in right eye (OD), 20/70 in left eye (OS), and the intraocular pressure (IOP) was 14 mmHg in OD and 20 mmHg in OS. Slit lamp examination of the OS revealed a corneal ulcer measuring 1.5mm horizontally and 6 mm vertically corresponding to the temporal clear corneal incision, a deep anterior chamber with 1+ flare and a posterior chamber implant in the capsular bag.
Treatment was started with topical fortified vancomycin 25 mg/mL and tobramycin 15 mg/mL hourly, scopolamine TID and oral moxifloxacin 400 mg daily. In the following days, the ulcer extended vertically to 7 mm, a hypopyon formed, a fibrin clot grew to fill the anterior chamber and the IOP rose to 24 mmHg. Corneal scrapings showed fungal elements and treatment was changed to oral voriconazole 200 mg BID, topical voriconazole 1% hourly, topical moxifloxacin 0.5% every 2 hours and continued with oral moxifloxacin. The vision in the OS dropped precipitously to counting fingers at 3ft on day 2 to hand motion on day 3. On day 4, the patient was hospitalized with an IOP of 41 mmHg, more fibrin and a larger hypopyon. Microbiology reported growth of methicillin resistant staphylococcus aureus(MRSA) sensitive to vancomycin and the treatment was changed to fortified vancomycin and voriconazole hourly, dorzolamide-timolol fixed combination BID, brimonidine TID, atropine and oral acetazolamide sequel 500 mg BID, voriconazole and moxifloxacin. Despite the intensive topical treatment and intravenous mannitol, the IOP fluctuated in the range of 40-57 mmHg. After informed consent, the patient agreed to the use of rTPA, which was prepared by the hospital pharmacy staff from lyophilized recombinant tPA (Activase, Genetech Inc, San Francisco). An anterior chamber injection of 0.1 ml rTPA (25 mcg/0.1 mL) preceded by aspiration of 0.08 ml of aqueous humor, was performed to clear the fibrin (Fig 1A). On postoperative day 4, slit lamp examination showed no change in the ulcer margins and a shallow anterior chamber with hyphema covering the hypopyon (Fig 1B). IOP in OS was 10 mmHg with only brimonidine TID. On postoperative day 15, the patient’s VA was CF at 1ft, slit lamp examination revealed a markedly retracted fibrin clot with traces of blood, no hyphema, minimal hypopyon and an IOP of 18 mmHg without glaucoma medication (Fig. 1C). At follow-up visits (week 3, week 5, week 8, week 12), the IOP had a favorable evolution (16, 15, 12, 31 and 18 mmHg, respectively) with brimonidine.
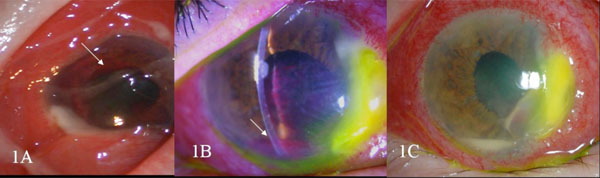
(A) Slit lamp photograph 2 hours post rTPA injection showing fibrin clot retraction superiorly (white arrow) and marked conjunctival congestion and chemosis. (B) Slit lamp photograph on day 4 post intracameral rTPA showing shallow anterior chamber, hyphema overlying the hypopyon (white arrow) and no progression of the corneal abscess. (C) Slit lamp photograph on day 15 post intracameral rTPA showing markedly reduced fibrin clot with minimal hypopyon.
DISCUSSION
Though acute inflammation within the eye may reduce aqueous secretion and lower the intraocular pressure, inflammatory material, consisting of white blood cells, macrophages and proteins, may obstruct the trabecular meshwork [5]. Additionally, inflammatory scarring may lead to peripheral anterior synechiae resulting in angle-closure glaucoma. In this case the secondary glaucoma was thought to be due to the inflammatory debris blocking the outflow of aqueous through the trabecular meshwork.
A surgical approach such as a trabeculectomy or tube shunt with an infectious corneal ulcer is not ideal for fear of intraocular extension of the infection, further intraocular inflammation or bleeding. A cyclodestructive procedure such as laser cyclophotocoagulation would encourage further inflammation and may lead to phthisis in this clinical setting. Therefore, the minimally invasive technique of rTPA injection became the favored initial approach to lower theIOP by dissolution of the fibrin clot. The effect of rTPA on the fibrin clot was seen in the next two hours but as a minor complication, a minimal hyphema covered the hypopyon. Nevertheless, the hyphema completely disappeared in 10 days and there was no further bleeding.
rTPA is a genetically cloned serine protease that works by converting plasminogen into plasmin. It promotes degradation of the fibrin only at the clot surface and it is not associated with systemic fibrinolysis. rTPA has been effectively used in the past in traumatic hyphema [1], fibrinous effusion after cataract surgery in adults [2], severe post cataract fibrinous membrane in pediatric groups [3], glaucoma surgery [4, 5 ] and subretinal hemorrhage [6]. The response to rTPA may have been coincident with an improving clinical course and unrelated to the drug. The potential complications related to the use of rTPA are corneal edema and anterior chamber turbidity, which results from immediate fibrinolysis products [5].
In conclusion, in markedly inflamed eyes with a fibrin blockade of aqueous outflow, the use of intracameral rTPA should be considered to lower IOP in eyes resistant to topical and systemic medications.


