All published articles of this journal are available on ScienceDirect.
NAVILAS Laser System Focal Laser Treatment for Diabetic Macular Edema - One Year Results of a Case Series
Abstract
Purpose:
To report one year outcomes of focal Navigated Retina Laser Therapy (NAVILAS) for diabetic macular edema (DME).
Methods:
Retrospective cohort series of 7 diabetic patients treated with NAVILAS focal laser. Statistical analysis included descriptive and continuous variables (Best-corrected logMAR Visual Acuity and time-domain optical coherence tomography (OCT) parameters) which were compared using a non-parametric procedure, the Friedman tests for repeated measures. A p-value of less than 0.05 was considered to denote statistical significance.
Results:
diabetic patients (4 male; 3 female) with an average age of 60.8 years (range 48-85 years) were included. All treated eyes were phakic; patients had an average hemoglobin A1C of 9.1 (range 7.8-11.7) at baseline and 8.0 (range 7.4-8.4) at 12 months. Six of the 7 patients had intravitreal bevacizumab injections prior to focal laser treatment with 1 patient having had more than 1 prior injection (total 3). At 12 months, median logMAR improved from 0.695 (± interquartile range 0.574) to 0.477 (± 0.573, p <0.001). OCT median central foveal thickness decreased from 248 (± 112) to 220 µm (± 41, p <0.001); total macular volume decreased from 7.84 (± 0.8) to 7.44 mm3 (± 0.7, p = 0.117); and largest macular subfield thickness decreased from 354 (± 116) to 289 µm (± 42, p <0.001). All patients were treated without complications.
Conclusions:
Focal NAVILAS showed to be safe and effective in treating DME with improvement in visual acuity and macular edema on OCT over 12 months in this case series. In clinical practice, combined treatment with focal laser including NAVILAS and anti-vascular endothelial growth factor may provide long-term improvement in DME.
introduction
Laser photocoagulation has been the standard of care for patients with clinically significant macular edema for several decades, but with several new advancements in treatment modalities including anti-vascular endothelial growth factor (VEGF), our ability to treat diabetic macular edema (DME) has significantly improved. The Early Treatment Diabetic Retinopathy Study (ETDRS) first showed the efficacy of focal laser therapy for treatment of DME [1], and now with the introduction of intravitreal anti-VEGF therapy with ranibizumab (Lucentis, Genentech Inc., San Francisco, CA, USA) and bevacizumab (Avastin, Genentech Inc., San Francisco, CA, USA) as seen in the ranibizumab for diabetic macular edema (RISE) and ranibizumab injection in subjects with clinically significant macular edema with center involvement secondary to diabetes mellitus (RIDE), prevention of further vision loss from macular edema has been substantial [2,3]. Although intravitreal anti-VEGF therapy has become one of the mainstays in treatment of DME, focal laser therapy remains important in treating extrafoveal DME, and individualized treatments with both modalities in clinical practice may become the future of DME care.
The ETDRS study as well as the Diabetic Retinopathy Clinical Research (DRCR) network have provided clinicians with guidelines for the treatment of DME, but there is little standardization or quality control when it comes to clinical practice. In addition, accuracy of the treatment is always a concern especially when applying focal laser treatment near the fovea. The navigated laser photocoagulator (NAVILAS; OD-OS GmbH, Teltow, Germany) is a new prototype of retinal eye-tracking laser delivery system with integrated digital fundus imaging (live color fundus photographs, red-free and infrared imaging, and fluorescein angiography) which could potentially increase the effectiveness and efficiency of focal laser therapy for DME by aligning pre-planned treatment maps with real-time fundus images. This scanning slit-lamp based system allows registered image overlay and laser stabilization on the retina using image processing in the treatment of retinal lesions [4-7]. The system uses a diode-pumped solid-state laser (532 nm) for both single-spot and pattern retinal photocoagulation [4]. Eye-tracking mechanisms of the instrument compensate for the eye’s excursions during treatment and utilize registration of retinal vessel positions and multiple retinal landmarks to limit the possibility of inadvertent placement of laser burns [4-7]. A contact lens can be used but is not necessary for accurate treatment. The physician can use either infrared or full color illumination to follow and modify the positioning of each marked laser spot as the system progresses during its treatment [6,7].
The aim of this study was to investigate the long-term results (12 months) of NAVILAS focal photocoagulation for a group of patients with poorly controlled diabetic macular edema who were also treated with anti-VEGF therapy.
Methods
New York University School of Medicine Institutional Review Board (IRB) approval was obtained for this Health Insurance Portability and Accountability Act-compliant, retrospective cohort study and all research adhered to the tenets of the Declaration of Helsinki (IRB #S12-00331). The medical records of 7 diabetic patients with clinically significant, diabetic macular edema treated with a navigated laser photocoagulator at the Bellevue Eye Clinic were reviewed. Patients were selected for focal laser treatment based on the presence of macular edema on OCT imaging and/or leakage on fluorescein angiography. Each treatment plan was individualized based on the number and size of microaneurysms seen on color fundus photography and fluorescein angiography. The treatments were performed without contact lens and without topical anesthesia. The laser parameters used in this study were a spot size of 100-150 µm, duration of 100-150 milliseconds (ms), and power between 50-180 milliwatts (mW).
Best-corrected logMAR (BCVA) and time-domain optical coherence tomography (OCT) (Stratus OCT, Carl Zeiss-Meditec Inc, Dublin, CA) parameters such as central foveal thickness (CFT), total macular volume (TMV), and largest subfield of macular thickness (LMT) were recorded at 3 month intervals over a 12 month follow-up.
Statistical analyses were carried out by one physician (A. L-P) and performed utilizing SPSS 21.0 (IBM Corp). The descriptive statistics are presented as median and interquartile range (IQR) values where available and measured ranges indicate minimums and maximums. Continuous variables for the entire cohort (BCVA and time-domain OCT parameters) were compared using a non-parametric procedure, the Friedman tests for repeated measures, after verifying that our sample was not normally distributed (by applying the Kolmogorov-Smirnov test) neither was there homogeneity in group variances (using the Levene`s robust test). A p-value of less than 0.05 was considered to denote statistical significance.
Results
We treated 7 diabetic patients (4 male; 3 female) with an average age of 60.8 years (± standard deviation 12.3, range 48-85 years). All eyes were phakic; patients had an average hemoglobin A1c of 9.1 (± 1.8, range 7.8-11.7) at baseline and 8.0 (± 0.5, range 7.4-8.4) at 12 months, and these differences were not significant (p=0.180) using the Wilcoxon Signed Ranks Test. Three patients were treated for hypertension and 1 patient showed history of hyperlipidemia. Average systolic blood pressure was 130.1 (± 16.7, range 115-159) and average diastolic blood pressure was 79.4 (±15, range 59-108). Six of the 7 eyes had intravitreal bevacizumab injections prior to focal laser treatment with 1 eye having had more than 1 prior injection (total 3). Two patients had a single treatment of manual focal laser prior to NAVILAS focal laser (Table 1). After 12 months of follow-up, 3 eyes required further intravitreal bevacizumab injections after NAVILAS focal laser treatment with each eye requiring 6, 10, and 3 injections at intervals of 34 days, 21 days, and 263 days after focal treatment, respectively. Average interval of required bevacizumab injection after NAVILAS treatment was 106 days (± 136.1, range 21-263). Table 1. Baseline Demographic and Clinical Characteristics of Patients Studied All laser applications hit the pre-planned microaneurysm points of focal leakage in all eyes (Fig. 1E, F). Color fundus photos and fluorescein angiography was performed before (Fig. 1A, C) and after (Fig. 1B, D) treatment and showed improvement in clinical features such as amount of hard exudate, number of leaking microaneurysms, and non-proliferative diabetic retinopathy. Average number of shots was 35.8 (± standard deviation 19.7, range 12-70); average amount of energy was 94.8 mW (± 26.2, range 50-146); average pulse duration was 99.2 ms (± 3.6, range 90-103); average spot size was 91.3 µm (± 29.8, range 50-140); and average total amount of energy administered to the eye during treatment was 2711.5 Joules (± 1896.2, range 714-5580). There were no complications or inadvertent photocoagulation of the fovea or optic nerve head.
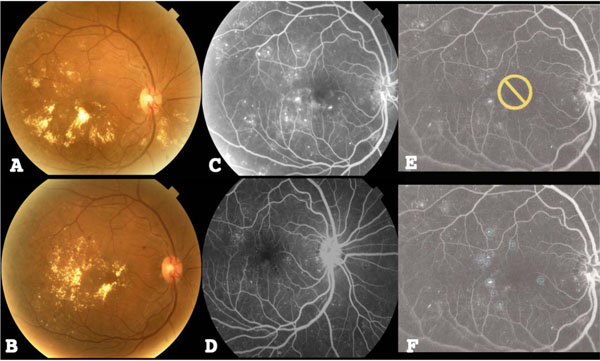
(A) Color fundus photo of the right eye of a study patient with DME before treatment. (B) Color fundus photo of the left eye after treatment. (C) Represents early fluorescein angiogram (FA) of the right eye prior to NAVILAS treatment. (D) Represents early (FA) of the right eye after treatment. (E, F) An image overlay composed and displayed over the pre-treatment FA showing planning elements of microaneurysm treatments and no-treatment zones.
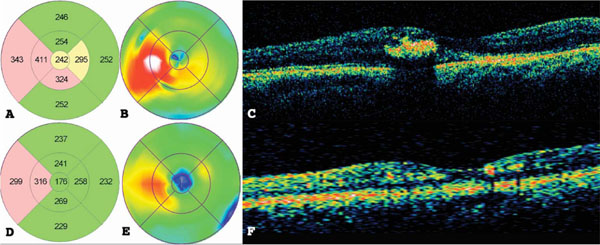
(A) Macular topographic map, (B) color macular map, and (C) macular cross-section optical coherence tomography (OCT) of the same study patient from Fig. (1) before treatment. (D) Macular topographic map, (E) color macular map, and (F) macular cross-section OCT after NAVILAS treatment.
Baseline Demographic and Clinical Characteristics of Patients Studied
| Demographic Characteristics | |
|---|---|
| Number of Patients | 7 |
| Age (mean) | 60.8 |
| Age (range) | 48-85 |
| # of OD | 4 |
| # of OS | 3 |
| HTN | 3 |
| HLD | 1 |
| Hgb A1c (mean) | 9.1 |
| Hgb A1c (range) | 7.8-11.7 |
| Systolic BP (mean) | 130.1 |
| Systolic BP (range) | 115-159 |
| Diastolic BP (mean) | 79.4 |
| Diastolic BP (range) | 59-108 |
| Prior Focal | 2 |
| Prior Bevacizumab | 6 |
Baseline demographic and clinical characteristics of patients included in the study were assessed. “#” = number. OD = right eye. OS= left eye. HTN = history of hypertension. HLD = history of hyperlipidemia. Hgb A1C = hemoglobin A1C at diagnosis. BP = blood pressure.
Analysis of Optical Coherence Tomography Parameters and Best-corrected LogMAR Visual Acuity
| BCVA LogMAR | OCT Central Foveal Thickness | OCT Macular Volume | OCT Largest Deviation | |
|---|---|---|---|---|
| Baseline | 0.695 ± 0.574 | 248 ± 112 | 7.84 ± 0.874 | 354 ± 116 |
| 3 months follow-up | 0.544 ± 0.447 | 258 ± 100 | 7.92 ± 0.630 | 354 ± 76 |
| 6 months follow-up | 0.397 ± 0.505 | 288 ± 163 | 7.88 ± 0.559 | 346 ± 93 |
| 9 months follow-up | 0.477 ± 0.367 | 288 ± 130 | 7.60 ± 1.315 | 312 ± 116 |
| 12 months follow-up | 0.477 ± 0.573 | 220 ± 41 | 7.44 ± 0.757 | 289 ± 42 |
Statistical median ± interquartile range of best corrected LogMAR visual acuity (BCVA LogMAR) and optical coherence tomography parameters (OCT) from baseline to each 3 month interval over 12 months in the entire sample.
Statistical Analysis of Means by Friedman Chi-Square Test
| BCVA LogMAR | OCT Central Foveal Thickness | OCT Total Macular Volume | OCT Largest Deviation | |
|---|---|---|---|---|
| Chi-Square value | 10.939 | 33.000 | 2.455 | 33.000 |
| Asymptotic significance | <0.001 | <0.001 | 0.117 | <0.001 |
Friedman repeated measures chi-square tests of best corrected LogMAR visual acuity (BCVA) and optical coherence tomography parameters (OCT) for the entire sample.
Before treatment, BCVA of all eyes showed a median (± IQR) of 0.695 ± 0.5, and OCT parameters were 248 ± 112 µm for CFT, 7.84 ± 0.8 mm3 for TMV and 354 ± 116 µm for LMT. At 12 months, comparing these variables to baseline, all participants’ BCVA improved to 0.477 ± 0.5 and OCT parameters: CFT decreased to 220 ± 41 µm; TMV decreased to 7.44 ± 0.7 mm3 and LMT decreased to 289 ± 42 µm, as can be observed in Table 2. These differences were statistically significant for BC LogMAR VA (p <0.001), OCT Central Foveal Thickness (p <0.001) and OCT Largest Deviation (p <0.001) and the ranks of these variables differ significantly from their expected value in the repeated measures with the Friedman chi-square tests, as can be observed in Table 3. An example of one patient’s OCT macular topographic map before (Fig. 2A, B) and macular cross-section OCT (Fig. 2C) and macular topographic map after (Fig. 2D, E) and macular cross-section OCT (Fig. 2F) showed a decrease in DME.
Discussion
Focal retinal laser remains an important primary treatment for DME. The ETDRS randomized 2244 patients with clinically significant macular edema (CSME) to either early treatment with focal and grid photocoagulation or deferral of treatment; and this landmark study showed at 3 years moderate visual loss was reduced by 50% with immediate focal argon photocoagulation treatment [1,3]. Visual acuity improved by more than 1 line in 40% of patients who underwent treatment compared with 22% in those who did not [1]. Diabetic macular edema occurs due to a malfunction of the blood-retinal barrier and death of endothelial cells leading to leakage of fluid and subsequent photoreceptor dysfunction [8]. Focal laser therapy leads to the occlusion of these leaking microaneurysms, pathologic vessels, or subretinal sites of leakage [6]. This closure is a delayed process that may extend up to 12 weeks after initial treatment [9]. As the ETDRS study showed, focal laser therapy can reduce moderate to severe vision loss, but the major effects were not seen till after 3 years of follow-up [1,3].
The NAVILAS system has been shown to have both a high degree of safety and combines the advantages of computerized precision and accuracy to efficiently perform focal retinal laser therapy [4-7,10,11]. Traditional focal laser therapy requires a contact lens, topical anesthesia, and the patient’s cooperation to fixate for the treatment period. Also, the ability of the retina specialist to locate each aneurysm based on the color fundus photograph or angiogram has the potential for treatment inaccuracy and often leads to physicians avoiding areas near the fovea, modifying their protocols and applying grid patterns [6], and performing several frequent retreatment sessions [12]. In comparison, retinal and eye-tracking systems such as the NAVILAS system align pre-planned treatment locations with the real-time fundus image increasing the potential effectiveness and efficiency [6,7]. Previously, it has been shown that a single focal therapy NAVILAS session can be efficiently completed including planning time in approximately 7 minutes and 47 seconds [4,7] and with significantly less pain compared to conventional laser treatment [10]. Similarly, all of our patients completed the NAVILAS treatment without complication and were also pleasantly surprised by the short duration of the procedure, the lack of discomfort, and the absence of a contact lens for the procedure.
Kozak et al. recently reported in June 2011 the accuracy of the NAVILAS system. In their analysis of the accuracy of 400 random focal targeted spots, they found that the system achieved a microaneurysm hit rate of 92% compared to the control group analyzing 100 focal spots performed manually, which was significantly lower at 72% [4]. Efficacy of NAVILAS focal laser treatment after 6 months showed statistically significant decrease in foveal thickness and improvement of visual acuity in 25 non-previously treated eyes with perifoveal telangiectasia (n=3), central serious chorioretinopathy (n=2), and diabetic macular edema (n=20) [6].
Patients in our study reflect an average population at a public hospital with poorly controlled diabetes. Frequently, the disease is less than optimal controlled and treatment success of any retinal disease is limited by hyperglycemia, increased blood pressure, and renal compromise. In our current study, we showed the continued effectiveness of NAVILAS focal therapy after 12 months of follow-up. Even with poorly controlled diabetes with average hemoglobin A1C before and after treatment of 9.1 and 8.0, respectively, we observed a statistically significant decrease in macular edema and improvement of best-corrected vision after 12 months. In our cohort, 6 of the 7 patients had been treated previously with intravitreal bevacizumab, but only 3 patients required an average of 6.3 more injections after NAVILAS treatment, ranging from 21 to 263 days after treatment. Whether this decrease in number of patients receiving injections was related to the NAVILAS focal treatment remains unclear, but it is possible that a combination of focal therapy and intravitreal anti-VEGF can improve the treatment of DME. These initial findings from study infer that there is a potential improvement with combination focal NAVILAS and intravitreal anti-VEGF in the treatment of DME, but larger randomized controlled studies are necessary to confirm these findings.
Our results agree with another recently published study by Neubauer et al. [13]. They concluded that navigated laser therapy enables a reduced retreatment rate and faster visual acuity gain compared with conventional slit-lamp based laser. Although their cohort of 46 eyes including a larger sample size compared to our study, they only observed patients that had focal NAVILAS alone whereas our results incorporate patients that have been treated with anti-VEGF. As recommended by recent studies including the ranibi-zumab mono-therapy or combined with laser versus laser monotherapy for diabetic macular edema (RESTORE) study, anti-VEGF treatment combined with focal laser has become a prominent first line treatment for DME [2,14]. Our results suggest that using focal NAVILAS in conjunction with intravitreal anti-VEGF bevacizumab can potentially maintain visual outcomes for patients with diabetic macular edema. Future larger studies utilizing this novel, efficient focal laser delivery system with anti-VEGF treatment and the number of required post-NAVILAS focal laser intravitreal injections may be helpful in expanding our ability to efficiently treat DME.
There are several limitations to our current study including an overall small number of patients and the fact that patients were not randomized to treatment. Given that our study was a retrospective case series, we recommend further studies including a randomized treatment trial with focal NAVILAS and intravitreal anti-VEGF to draw definitive conclusions.
Focal retinal laser remains a long-term stabilization treatment for DME and is a proven therapy based on the initial ETDRS study. In this clinical series with 12 months of follow-up, we suggest that NAVILAS focal laser can be a safe and accurate alternative form of treatment for diabetic macular edema. Even with the development and increased usage of the intravitreal anti-VEGF therapy for macular edema, there still remains a role for focal. Combining these two modalities may increase the effectiveness of treatment and possibly lessen the long-term burden of continuous intravitreal anti-VEGF treatments.
Conflict of Interest
No financial contributions to the work being reported or potential conflicts of interest.
Financial Disclosures include:
Dr. Jesse J. Jung - None.
Dr. Roberto Gallego-Pinazo - Novartis Pharmaceuticals Corporation (research funding, consultancy services, serving on speakers’ bureaus, travel expenses); Alcon Laboratories, Inc. (travel expenses); Allergan (research funding); Bayer (research funding, consultancy services, travel expenses); Carl Zeiss Meditec (consultancy services, travel expenses)
Dr. Jonathan I. Huz - None.
Dr. Irene A. Barbazetto - None.
Dr. Antonio Lleó-Pérez - None.
Acknowledgements
This work was supported by The Macula Foundation, Inc.


