All published articles of this journal are available on ScienceDirect.
Inter-changeability Between Anterior Segment Optical Coherence Tomography and Scheimpflug Imaging in the Evaluation of Corneal Opacity
Abstract
Purpose
To compare the results of corneal evaluation, either the total corneal thickness and corneal opacity depth by both technologies AS- OCT and Scheimpflug imaging by Sirius Scheimpflug placido topography.
Methods
sixty-eight eyes of sixty- eight consecutive patients with corneal opacities were examined by both AS-OCT and Sirius Scheimpflug imaging. Corneal thickness and corneal opacity depth were measured and compared determining the interchangeability using Bland–Altman plotting.
Results
Total corneal thickness was (470.32±62.10) and (463.47±56.47) by AS. OCT and Sirius Scheimpflug imaging, respectively (P value = 0.8).
While the actual measurement of corneal opacity, corneal opacity depth was (135.39±119) and (85.45±128) by AS-OCT and Siris Scheimpflug imaging respectively with P value (0.004).
Conclusion
AS-OCT and Scheimpflug imaging Sirius may be used interchangeably for measuring both total corneal thickness, while in the determination of corneal opacity depth. the AS-OCT is superior to Scheimpflug imaging in the accuracy of determination of actual boundaries of the corneal opacity.
1. INTRODUCTION
Anterior segment imaging has shown many advances in the last years. The new imaging devices currently give accurate measurements of corneal power, corneal elevation, iridocorneal angle, anterior segment biometry, corneal aberrometry, and corneal pachymetry [1].
Accurate Corneal thickness measurement is mandatory in preoperative corneal evaluation, especially when performing corneal refractive surgery such as laser in situ keratomileusis, photorefractive keratectomy as the procedure of choice and the degree of correction depend mainly on the corneal thickness [2].
Various noncontact imaging technologies are available for corneal thickness measurement including interferometry [3], Optical Coherence Tomography (OCT) [4] and Scheimpflug camera [5]. These noncontact instruments have good repeatability in normal eyes [6, 7].
Optical coherence tomography was improved for anterior segment applications with higher wavelength imaging and faster image acquisition (0.125 seconds) using the same principles of reflection as for US pachymetry [5]. Remarkable advantages over alternative noncontact techniques include its accuracy even in the presence of corneal opacity, simple use, and high-resolution images [1].
The Scheimpflug camera (SC) combined with Placido corneal topography (Sirius; CSO Inc, Firenze, Italy) is a topography device with the ability of speedy acquisition of cross-sectional images of the cornea. It can measure about 35,632 points on the anterior corneal surface and 30,000 on the posterior corneal surface in high-resolution mode, in approximately 5-6 s. A detailed pachymetry is produced by using the point-by-point anterior and posterior corneal surface data [5].
Optical coherence tomography (OCT) has been used to evaluate the histopathology of corneal opacities [8]. Corneal haze could limit the light transmission resulting in backward and forward scattering. Light-backscattering analysis with OCT was found to be a reliable method of detecting backscatter [9, 10].
Scheimpflug photography of the anterior segment is relatively new imaging technique and superior to slit-lamp photography. In comparison with anterior segment optical coherence tomography, it provides a better evaluation of light scattering and can assess different degrees of corneal infiltrates [10].
Corneal optical clarity is essential for normal ocular function, Scheimpflug-based optical densitometry of the cornea is considered a new imaging method for corneal opacity evaluation [11].
The aim of our study was to compare the results of corneal evaluation of either the total corneal thickness and corneal opacity depth by both technologies AS- OCT and Scheimpflug imaging by Sirius Scheimpflug placido topography.
2. PATIENTS AND METHODS
This study is a cross-sectional study that included sixty-eight patients with different degrees of corneal opacifications who presented to the Sohag Investigative Ophthalmic Center in the period between July 2020 to April 2021.
All patients involved in this study were informed about the aim of the study and signed the informed consent. Our study adhered to the tenants of Helsinki and the ethical board committee approval of our institution (Sohag Faculty of Medicine) was obtained under IBR Registration number: Soh-Med-33-12-30.
Complete ophthalmic examination was performed in all patients, including visual acuity assessment, manifest refraction, slit lamp biomicroscopy, and fundus examination if possible.
All eyes included in this study were photographed by two devices; the first was the spectral-domain Optovue anterior segment OCT system, which uses a beam of infrared light of 840 nm wavelength to scan all structures of the anterior segment [12, 13]. The OCT system is connected to a computer with special software which provides different image capturing and measurement modalities. The incorporated software eliminates image distortion in the measurements induced by optical factors of transmission and provides different anatomic information. Both the thickness of opacity and total corneal thickness was measured in microns. The thickness of the opacity was determined by the deepest point of the opacity in the corneal stroma. The total corneal thickness over the opacity (which is defined as the total corneal thickness from the epithelium to endothelium over the deepest point of the opacity measured previously) and the location of the opacity were variable in the cases to be central, paracentral or in the mid-peripheral. The central corneal thickness was not the point of interest to compare so it was not measured.
In AS-OCT examinations, patients were asked to sit in front of the device with the head well positioned on the chin-and-forehead rest, looking at the center of an internal-fixation target. External fixation was used if the patient had vision impairment in the examined eye. The operator adjusted the infrared light on the center of the cornea and the pathological tissue and the noise level to obtain a clear image.
A vertical flare in the central-corneal zone enabled the identification of the corneal vertex.
Depending on the location of corneal opacities, a horizontal or vertical scan line was chosen for analysis. Moreover, the deepest point of the corneal opacity in the stroma was chosen to be measured by the caliper.
The boundaries of the corneal opacity were determined by the demarcation line between the hyper reflective stromal tissue and the clear corneal rim.
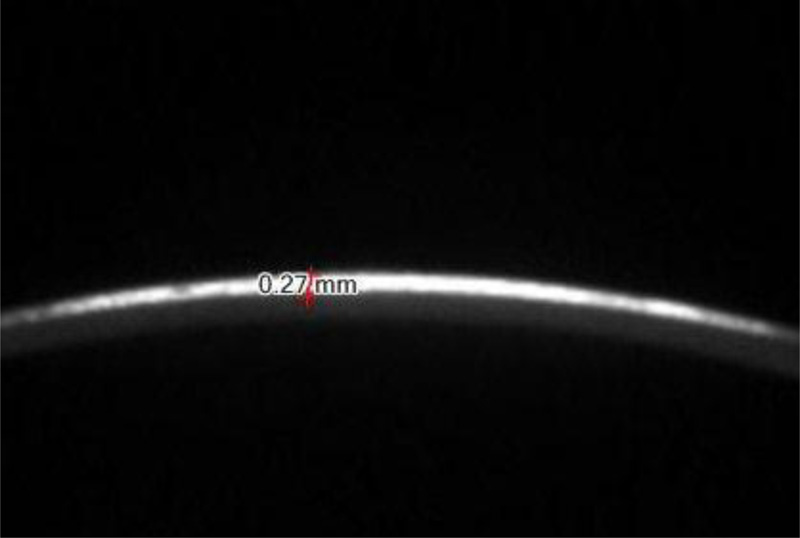
The second investigation was Scheimpflug-based optical densitometry of the cornea having been done for the patient using Sirius Scheimpflug placido topography (CSO, Florence, Italy). The depth of opacity and the total corneal thickness were measured by the caliper tool in the device software in mm which was later converted to microns (Fig. 1). The CSO topography system analyzes a total of 6144 corneal points of a corneal area included in a circular annulus outlined by an inner radius of 0.33 and an outer radius of 10 mm with respect to the corneal vertex.
Mesopic pupil diameter was acquired in a semi-dark room with the disc illuminated in a manner to bring the ambient light intensity to 4.0lux as advised by the manufacturer [14]. The patient's eye was aligned along the visual axis using a central fixation light. Patients were asked to blink between acquisitions to ensure good tear film.
Both examinations were done twice by the same experienced operator to ensure accuracy. the mean of both measures in each device was calculated and used as a net result for both devices and variables
All measurements by both devices were taken on the same day for each case between 10 am to 1 pm to avoid changes in measurement resulting from diurnal variation.
This research received no grant from any funding agency in the public or commercial sectors.
2.1. Statistical Analysis
Data were analyzed using MedCalc version 18.1. Quantitative data were represented as mean, standard deviation, median, and range. Differences between the instruments were compared to zero difference using the t-test, and Bland-Altman graphs were used to show the measurement differences of mean values. Bland-Altman graphs were also used to assess the agreement of measurements between the two devices. A value of p < 0.05 was considered statistically significant.
3. RESULTS
The clinical features of the study population are listed in Table 1. Patients’ age ranged from 13-54 years (mean =31.868±9.56 years) with a male to 89% to 42.11%), the total corneal power and corneal power over opacity are shown in Table 1.
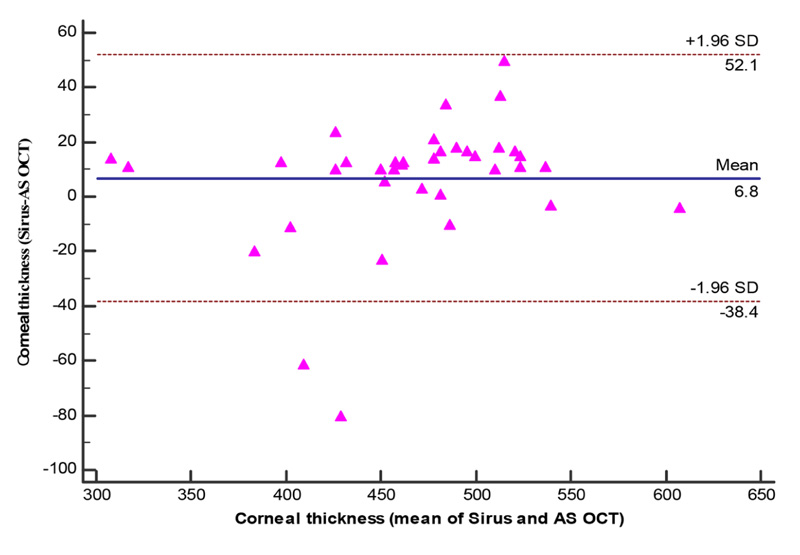
The total corneal thickness ranges between 301 um to 609 um by AS-OCT with a mean ±SD (463.47±56.47um), however by Sirius it ranges between 315 um to 605 um with a mean ± SD of 470.32±62.10um and the difference being statistically insignificant (P=0.08). Fig. (2) while the corneal opacity thickness differs with AS-OCT than Sirius Scheimpflug imaging as it ranged between 31 um to 382 um with a mean±SD of 135.39±119.19 um .
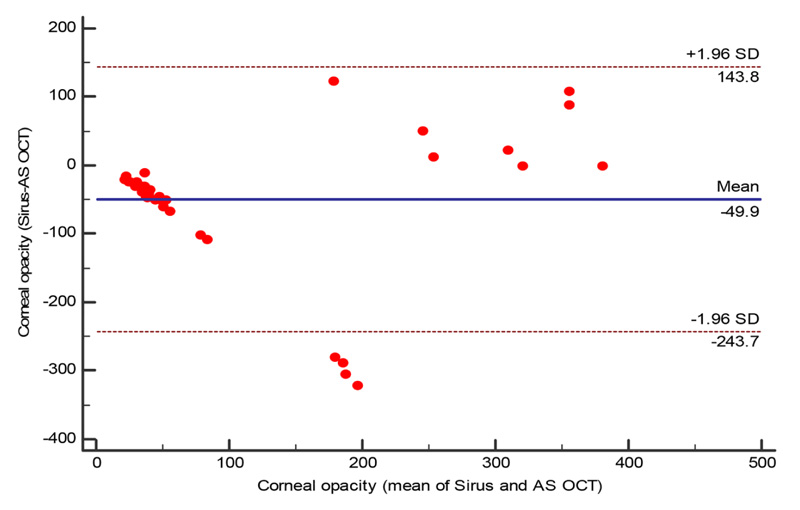
In Sirius Scheimpflug imaging it ranged between 11 to 409 um with a mean±SD 85.45±128.73 um and the difference is statistically significant (p=0.04) Table 2 and Figs. (3, 4).
| Variable | Summary Statistics |
|---|---|
|
Age/ years Mean ±SD Median (range) |
31.87±9.56 33 (13:54) |
|
Gender Females Males |
16 (42.11%) 22 (57.89%) |
|
K1 Mean ±SD Median (range) |
42.81±4.36 43.81 (20.84:50.66) |
|
K2 Mean ±SD Median (range) |
46.09±6.23 45.97 (24.78:72.13) |
|
K max over opacity Mean ±SD Median (range) |
53.65±10.45 50.36 (43.17:104.9) |
|
K min over opacity Mean ±SD Median (range) |
48.65±7.27 48.13 (26.17:81) |
| Parameter | Sirus | AS OCT | Difference | P Value |
|---|---|---|---|---|
|
Total Corneal thickness Mean ±SD Median (range) |
470.32±62.10 481 (315:605) |
463.47±56.47 469.5 (301:609) |
6.84±23.10 -38.4:52.1* |
0.08 |
|
Corneal opacity thickness Mean ±SD Median (range) |
85.45±128.73 22 (11:409) |
135.39±119.19 71 (31:382) |
-49.94±98.88 -243.7:143.8* |
0.004 |

4. DISCUSSION
The cornea is an important part of the optical refractive system and contributes to the powerful optical part of the eye as two-thirds of the total optical power is within the cornea [12].
It is well known that the corneal thickness in a healthy cornea range from 500–800µm and has a radius of 11–12mm [12].
The importance of corneal thickness measurement is needed in the diagnosis of several corneal disorders, including corneal degeneration, corneal dystrophy, and endothelial pump failure [15].
In addition, accurate evaluation of the corneal thickness is mandatory before any corneal surgery or maneuver, and corneal opacity depth measurement is needed to determine the best option for management and follow-up, many anterior segment devices with different technologies are present in the field nowadays to measure the corneal thickness, however fewer options of imaging technologies to evaluate and measure the corneal opacity. The most important anterior segment technologies we need to compare to judge interchangeability are the anterior segment OCT and Scheimpflug imaging.
It was known that the optical coherence tomography accurately maps corneal thickness in clear and opacified corneas, allowing the examiner to precisely map the depth of corneal opacities, the degree of epithelial hyperplasia and the thickness of the cornea [16]. Optical coherence tomography imaging is based on measuring the delay of light (typically infrared) reflected from tissue structures [17].
The OCT is superior to other techniques using a similar principle due to higher sensitivity and photodetection at the interferometer output involving multiplying the two optical waves together. Therefore, the weak signal in the object arm, backscattered or transmitted through the tissue, is amplified by the strong signal in the reference arm. This characteristic explains how OCT can typically detect reflected signals as small as one part in 10−10 of the incident power [17].
The Scheimpflug imaging system can provide cross-sectional images of the whole anterior segment with a larger depth of focus and better spatial accuracy than the Scanning-slit system, commonly using a traditional camera containing a coaxial optical path.
The rotating Scheimpflug imaging systems provide many advantages over the scanning slit systems. The most important advantage is the precise representation of the posterior corneal surface individually to ensure the accuracy of pachymetry measurements both in the center and in the periphery, especially in postoperative eyes where the curvature ratio between the anterior and posterior surfaces is changed [18].
In this study, there were sixty-eight eyes with corneal opacity, the mean age of the patients was 31.87±9.56 years, as most of the patients were in middle age (median 33 years), and the incidence of corneal opacities due to variable causes is frequent in this age regarding post traumatic, post inflammatory, and corneal dystrophies, while in another previous study [19] which studied evaluation of congenital corneal opacity in children ranging from 2 days to 2.5 years, actually the cause of the opacity was not studied as the evaluation of the established corneal opacity is the aim.
In this study, the total corneal thickness over opacity was measured by two devices, the results were quite the same with a difference of no statistically significant, which concludes that the AS- OCT and Sirius Scheimpflug imaging are comparable and any of them give accurate results about the total corneal thickness and each of both technologies can be used safely before any corneal surgery, this highly agreed with the study done by Huang et al. who found that the results of Scheimpflug imaging and FD Oct are repeatable and reproducible measurement for central corneal thickness and midperipheral corneal thickness [20]. While another study recommended that the two technologies should not consider interchangeable in measuring the corneal thickness as the corneal thickness was higher in anterior segment OCT [21].
Moreover, we found that our study gave the same conclusion about the interchangeability between the two devices as proved before by the study done by Chen at al [7]. While Kumar found a difference between both technologies which was statistically significant. He claimed that both devices should not be used interchangeable due to low agreement regarding central corneal thickness [22].
The AS-OCT is superior than the Scheimpflug imaging in mapping the structure of the cornea in details, giving the accurate thickness of the epithelium and the stromal thickness, as the OCT accurately maps the corneal thickness in clear and opacified corneas, allowing the examiner to precisely measure the depth of corneal opacities [16].
This agrees with Chan et al. who mentioned that AS-OCT is better in detecting central corneal thickness in Keratoconic eyes and he recommended Swept-source OCT might be preferred over Placido-Scheimpflug imaging owing to better repeatability of measurements [23].
While in normal healthy corneas, there was an agreement between the two devices in measuring the central corneal thickness which can be used interchangeably as mentioned by the study of Chen et al. [7].
While other reports as Ashour et al. found that OCT measurements are higher than those of Pentacam in most of the cases regarding CCT. In keratoconic and scarred cornea [24] and in a previous study comparing the central corneal thickness in an edematous cornea, it was found that Pentacm schiempflug imaging overestimates the thickness over the visante anterior segment OCT and Cirus OCT [25].
In this study, we found that the corneal opacity depth was different in the anterior segment OCT measurement and Scheimpflug imaging by Sirius, and this difference was statistically significant, giving the AS-OCT the preference for accurate measurement of corneal opacity. The advances in the technology of AS-OCT regarding the increased image acquisition speed and resolution allow the clear delineation and mapping of the corneal layers with any pathological changes in them [26].
AS-OCT was used accurately in the evaluation of postinfectious corneal opacities and envaulted their response to medical treatment to reduce scarring [27]. It also has the ability to classify the corneal opacitiesaccording to their severity by measuring the average pixel intensity of the corneal transparency [28]. It also provided an excellent evaluation of the depth, extent, and affection of different corneal layers in corneal dystrophies in detail [29, 30].
While Peymen and Peuriziz found that Scheimpflug-based optical densitometry of the cornea is able to objectively measure corneal haziness in infectious keratitis through a quantitative numerical assessment of the backscattered light [11], but no study had compared the two technologies in measuring the corneal opacity thickness before.
One of the limitations of this study is the lack of opacity grading by both devices which may be studied later, as the study aimed to judge if both devices could be used interchangeably and to detect which device would offer a more efficient estimation of corneal opacity thickness.
CONCLUSION
AS-OCT and Scheimpflug imaging Sirius may be used interchangeably for measuring both total corneal thickness, while in the determination of corneal opacity depth the AS-OCT is superior to Scheimpflug imaging in the accuracy of determination of actual boundaries of the corneal opacity.
LIST OF ABBREVIATION
| SC | = Scheimpflug Camera |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The ethical board committee approval of our institution (Sohag Faculty of Medicine) was obtained under IBR Registration number: Soh-Med-33-12-30.
HUMAN AND ANIMAL RIGHTS
No animals were used for studies that are the basis of this research. All the humans were used in accordance with the Helsinki Declaration of 1975.
CONSENT FOR PUBLICATION
All patients involved in this study were informed about the aim of the study and signed the informed consent.
STANDARDS OF REPORTING
STROBE guidelines were followed.
AVAILABILITY OF DATA AND MATERIALS
The excel sheet data used to support the results of this study are available from the corresponding author upon request [A.M].
FUNDING
None.
CONFLICT OF INTEREST
The author declares no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Declared none.


