All published articles of this journal are available on ScienceDirect.
Perifoveal Microcirculation in Macular Oedema with Retinal Vein Occlusion
TO THE EDITOR
Central retinal vein occlusion (CRVO) is a common retinal vascular disease that often results in macular oedema, which is the most frequent cause of visual impairment in these patients [1]. Therefore, it is important to understand the vascular and haemodynamic abnormalities that underlie the progression of macular oedema in CRVO. Remky et al. [2] found that the perifoveal blood flow velocity (BFV) was significantly reduced in CRVO patients compared with healthy subjects by using scanning laser ophthalmoscopy with fluorescein angiography (FA) to measure BFV in the perifoveal capillaries based on the movement of fluorescent dots. However, the relationship between perifoveal BFV and macular oedema in CRVO patients remains unclear. Accordingly, we investigated the relation between perifoveal capillary BFV and retinal thickness at the central fovea in CRVO patients with macular oedema, healthy volunteers, and patients who had branch retinal vein occlusion (BRVO) with macular oedema [3].
Seven CRVO patients with macular oedema were studied, including 4 men and 3 women with a mean age of 65.0 ± 4.9 years (Table 1). The fundus findings were confirmed by standardized fundus color photography and FA, which was performed with a Topcon TRC-50EX fundus camera, an image-net system (Tokyo Optical Co. Ltd., Japan), and a preset lens with a slit-lamp. Perifoveal BFV was measured by FA combined with scanning laser ophthalmoscopy and the tracing method [3]. Retinal thickness at the central fovea was measured by optical coherence tomography with a Zeiss-Humphrey apparatus (Zeiss-Humphrey Ophthalmic Systems, Dublin, California, USA). Each subject underwent complete ophthalmic evaluation. The data obtained were compared with findings previously reported for patients with BRVO and healthy volunteers [3]. Results are expressed as the mean ± standard error and the range. To compare BFV data among the groups, ANOVA and a Tukey-Kramer post hoc test were performed. To examine correlations between parameters, Spearman’s rank correlation coefficients were calculated by linear regression analysis. A two-tailed P value of less than 0.05 was considered to indicate statistical significance. This study was conducted according to the tenets of the Declaration of Helsinki, and was approved by the institutional review board. All subjects provided informed consent.
Profile of the Patients With Central Retinal Vein Occlusion
| Patient No. | Age (y) Gender | Duration of CRVO (Months) | Hypertension | Retinal Thickness (µm) | BFV (mm/sec) |
|---|---|---|---|---|---|
| 1 | 60 F | 5 | + | 583 | 0.80 |
| 2 | 58 M | 3 | + | 623 | 0.77 |
| 3 | 63 M | 4 | - | 470 | 0.84 |
| 4 | 67 F | 3 | + | 835 | 0.45 |
| 5 | 66 M | 5 | + | 714 | 0.73 |
| 6 | 70 M | 7 | + | 512 | 1.10 |
| 7 | 71 F | 5 | + | 611 | 0.79 |
CRVO = central retinal vein occlusion; BFV =perifoveal capillary blood flow velocity.
As shown in the Fig. (1), the mean perifoveal capillary BFV decreased significantly across the three groups of subjects from healthy controls (1.49 ± 0.11 mm/sec) to BRVO patients (1.08 ± 0.28 mm/sec) and then CRVO patients (0.78 ± 0.19 mm/sec) (p<0.001). There was a significant negative correlation between perifoveal capillary BFV and retinal thickness at the central fovea in the CRVO patients (r=–0.85, p=0.014).
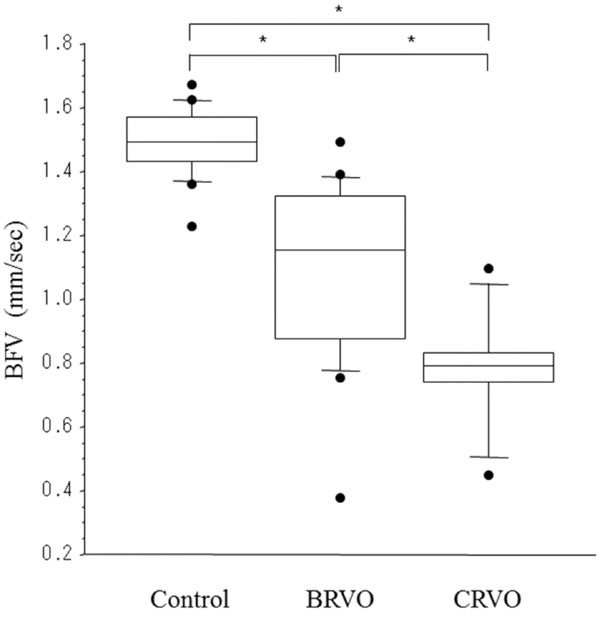
Capillary blood flow velocity (BFV) in healthy controls (n=16) (3), BRVO patients (n=18) (3), and CRVO patients (n=7). BFV decreased significantly across the three groups (healthy controls: 1.49 ± 0.11 mm/sec, BRVO patients: 1.08 ± 0.28 mm/sec, CRVO patients: 0.78 ± 0.19 mm/sec) (p<0.001). The top and bottom ends of the box indicate the 25 and 75 percentiles, line in the box indicates the median, and the top and bottom ends of the error bars indicate the 2.5 and 97.5 percentiles. * p<0.05 by the Tukey-Kramer multiple comparison method.
This study showed that the perifoveal capillary BFV was significantly lower in patients with CRVO than in healthy persons and BRVO patients. This difference may have been detected because functional and/or anatomic capillary closure is more advanced in CRVO. It is possible that a reduction of BFV in CRVO leads to functional vascular obstruction and relative retinal ischaemia, resulting in the increased production of molecules like vascular endothelial growth factor (VEGF), which potently increases vascular permeability [4]. VEGF also increases the expression of intercellular adhesion molecule-1 (ICAM-1) by cultured endothelial cells, leading to the rolling, adhesion, and migration of leukocytes [5]. Accordingly, it can be suggested that chronic trapping of leukocytes in the capillaries of CRVO patients may lead to impaired perfusion, resulting in the overexpression of VEGF and ICAM-1 along with the development/progression of macular oedema. Thus, VEGF and ICAM-1 may both play a role in reducing the BFV of CRVO patients with macular oedema.
In conclusion, a decrease of perifoveal capillary BFV may have a role in the pathogenesis of macular oedema in patients with CRVO.
ACKNOWLEDGEMENT
Declared none.
CONFLICT OF INTEREST
Declared none.
PROPRIETARY INTEREST
The authors do not have any proprietary interest in this manuscript.
GRANTS AND FUNDS
The authors have not received any financial support.


