All published articles of this journal are available on ScienceDirect.
Evaluation of Pollen Adhesion to Verofilcon-A Soft Contact Lenses
Abstract
Purpose:
A new 1-day disposable soft contact lens (SCL), verofilcon-A, constructed of silicone hydrogel material, has recently become available in Japan. This SCL has a very smooth surface produced by using the SMARTSURFACE ® Technology, and it was expected that pollen particles and protein components would not adhere easily to its surface. We examined the degree of pollen adhesion to the surface of the verofilcon-A material SCL and compared the results with those of other 1-day disposable SCLs (1DSCL).
Methods:
To determine the number of pollen grains attached to the surface of different types of SCLs, 0.01 mg/ml of cedar pollen solution was dropped onto the surface of 13 types of 1DSCL. After 24 h, each 1DSCL was rinsed in a shaker and washed five times with saline (n = 10 for each 1DSCL type). The number of pollen particles adhered to the 1DSCL and the percentage of surface area occupied by pollen was determined.
Results:
The number of pollen particles on the 1DSCLs ranged from 0 to 185 in the 200 × 200 µm area. The number of particles was lowest in the delefilcon-A and verofilcon-A SCLs with 0 particles, and the number was higher in the other 11 1DSCLs. The number of pollen particles was negatively correlated with the water content (r = −0.48), oxygen permeability (Dk; r = −0.43), oxygen transmissibility (r = −0.42), and center thickness (r = −0.33) of the 1DSCLs. The pollen adhesion area ranged from 0.0% to 3.1% and was lowest in the delefilcon-A and verofilcon-A 1DSCLs. There were significant differences in the pollen adhesion area between colored 1DSCLs (2.73 ± 1.97%) and clear 1DSCLs (1.03 ± 1.01%, P<0.001) and between hydroxyethyl methacrylate-based 1DSCLs (1.84 ± 1.45%) and silicone hydrogel-based 1DSCLs (0.05 ± 0.16%, P<0.001).
Conclusion:
These findings indicate that the verofilcon-A 1DSCL processed with SMARTSURFACE™ Technology is an excellent option for SCL users with allergic conjunctivitis during the high pollen season.
1. INTRODUCTION
Seasonal allergic conjunctivitis is usually caused by airborne allergens, such as the pollen of trees, grasses, and weeds, and worsens during the high pollen season. The typical signs of seasonal allergic conjunctivitis are mild to moderate itching, redness, and swelling of the conjunctiva [1, 2]. Soft contact lenses (SCLs) also cause giant papillary conjunctivitis (GPC), a subtype of allergic conjunctivitis. However, contact lens-induced GPC is not real allergic conjunctivitis, but is considered a chronic ocular inflammation [1, 3]. During wear, the SCLs accumulate tear film deposits3 including proteins [4, 5], lipids [6], and mucins [7]. The Food and Drug Administration Groups II and IV SCLs are high water content SCLs that accumulate more tear film components than Groups I and III low water content SCLs [5, 6]. The SCLs can easily trap various aeroallergens including tree and grass pollen, dust, smog, and cosmetic makeup. These allergens worsen the symptoms of allergic conjunctivitis and may cause conjunctival inflammation [8-10].
An earlier study suggested that the rate of adhesion Cryj 1, a major Japanese cedar pollen allergen, to SCLs was higher in the monthly replacement SCLs than the daily disposable SCLs or the 2-week frequent replacement SCLs [11]. Another study demonstrated that the pollen adhesion to daily disposable SCLs was higher on the colored SCLs than on the clear SCLs [12]. Additionally, pollen adhesion was lower in the silicone hydrogel SCL than in hydroxyethyl methacrylate (HEMA) CLs [12]. These results suggest that the daily disposable silicone hydrogel type of SCL may be better for patients with AC, especially during the pollen season.
Recently, a new 1-day disposable SCL (1DSCL) made of silicone hydrogel called verofilcon-A (PRECISION1™, Alcon Japan Ltd. Tokyo, Japan) was introduced to Japan. Verofilcon-A is made from a new high oxygen permeability (Dk; 90 × 10−11 barriers) material with a 2–3-µm-thick surface with more than 80% water content and is a Class 1 ultraviolet blocker (≥ 90% of UVA, ≥ 99% of UVB) [13]. The verofilcon-A SCL has a smooth surface with the smoothing produced by SMARTSURFACE® Technology, and it is expected that pollen and protein components will not adhere to this type of surface as easily. However, whether there is less pollen adherence to verofilcon-A SCLs than other types of 1DSCLs has not been determined.
We examined the degree of pollen adhesion to the surface of verofilcon-A SCLs and compared the degree of pollen adhesion to verofilcon-A daily disposable SCLs to that of other major 1DSCLs available in Japan.
2. MATERIALS AND METHODS
2.1. Research Design
This study performed a nonclinical and comparative analysis. This study was reviewed and approved by the Ethics Committee of Teikyo University.
Thirteen different types of −4.0 diopter 1DSCL were tested (n = 10 each) (Table 1). A total of 130 1DSCLs were used in this study. Japanese cedar pollen (Cryptomeria japonica) was purchased from the Yamizo Pollen Study Group (Daigo-cho, Ibaraki Prefecture, Japan). According to the manufacturer’s instructions, pollen was collected from naturally dried male cedar flowers and purified using a filter.
2.2. Adhesion of Pollen to SCLs
0.2 ml phosphate-buffered saline (PBS) containing 0.02 mg cedar pollen was dropped onto the surface of 1DSCLs. After that, SCLs were maintained at room temperature for 24 h [14]. The SCL was then placed in a dish containing 10.0 ml of PBS and shaken five times for 1 min to remove the pollen from the surface of the SCLs. The SCL was rinsed three times in PBS. The central part of the SCL was photographed using a microscope. The number of pollen particles adhering to a 200 µm × 200 µm area in the central part of the SCL and the portion of the adhering area were calculated using ImageJ analysis software (version 1.52a).
2.3. Statistical Analyses
Two-tailed unpaired Student’s t-tests were used to determine the significance of the differences in the mean number of pollens among the 13 groups of SCLs. The mean values among three or more groups were compared by one-way analysis of variance (ANOVA) using the Tukey-Kramer method and the Kruskal-Wallis test. Correlation analysis was performed using two-tailed Pearson correlation coefficients. Factors affecting the adhesion of pollen to SCLs were investigated using multivariate logistic regression analyses. Data are expressed as mean ± standard deviation or percentage. If the p-value is 0.05 or lower, the result is considered significant.
3. RESULTS
3.1. Numbers of Pollen Particles Adherent to SCLs
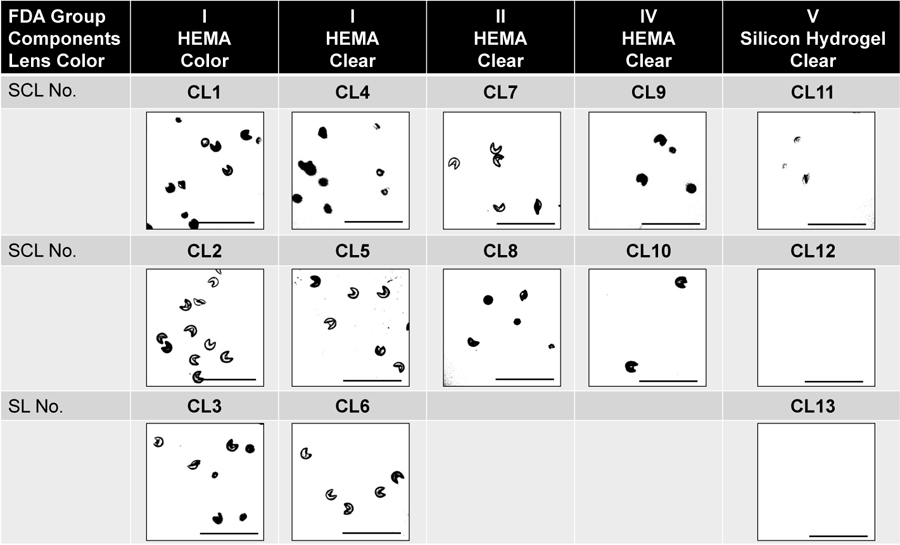
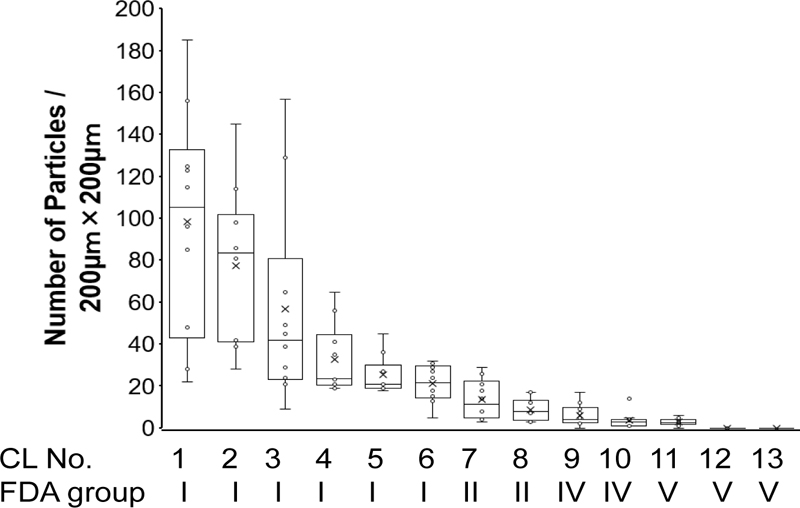
Fig. (1) shows the pollen particles attached to the surface of the SCLs/200 μm × 200 μm area. The number of pollen particles adhering to the SCL was 98 ± 51 for CL1, 77 ± 36 for CL2, 57 ± 46 for CL3, 33 ± 16 for CL4, 25 ± 8 for CL5, 21 ± 8 for CL6, 14 ± 9 for CL7, 9 ± 5 for CL8, 6 ± 5 for CL9, 4 ± 4 for CL10, 3 ± 2 for CL11, 0 ± 0 for CL12, and 0 ± 0 for CL13 (Fig. 2). There were significant differences among the 13 groups (P<0.001).
| SCL No. |
Water Content (%) | Oxygen Permeability (Dk) |
Oxygen Transmissibility (Dk/L) |
Diameter (mm) |
Base Curve (mm) |
CT (mm) |
Colored SCL |
Surface (Ionic/ Non-ionic) |
FDA group |
USAN Nomenclature |
Principal Components |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 38.6 | 8.5 | 12.1 | 14.2 | 8.7 | 0.08 | Yes | Non-ionic | I | Polymacon | 2-HEMA, EGDMA |
| 2 | 38 | 12 | 24 | 14.5 | 8.7 | 0.05 | Yes | Non-ionic | I | Polymacon | HEMA, EGDMA |
| 3 | 42.5 | 11 | 13.75 | 14 | 8.6 | 0.08 | Yes | Non-ionic | I | Polymacon | HEMA、NVP、MMA |
| 4 | 38.5 | 10 | 14.3 | 14 | 8.7 | 0.07 | No | Non-ionic | I | Polymacon | HEMA, EGDMA |
| 5 | 38.5 | 10 | 20 | 14 | 8.7 | 0.05 | No | Non-ionic | I | Polymacon | HEMA, EGDMA |
| 6 | 38 | 8.5 | 12.1 | 14 | 8.7 | 0.07 | No | Non-ionic | I | Polymacon | HEMA, EGDMA |
| 7 | 58 | 25.68 | 36.7 | 14.2 | 8.6 | 0.07 | No | Non-ionic | II | Omafilcon A | 2-HEMA |
| 8 | 59 | 22 | 24.2 | 14.2 | 8.6 | 0.09 | No | Non-ionic | II | Hilafilcon-B | HEMA, NVP |
| 9 | 58 | 28 | 33.3 | 14.2 | 8.75 | 0.084 | No | Ionic | IV | Etafilcon-A | HEMA, MA |
| 10 | 55 | 19.7 | 26.3 | 14.2 | 8.6 | 0.075 | No | Ionic | IV | Ocufilcon-D | HEMA, MA |
| 11 | 38 | 103 | 121 | 14.3 | 8.75 | 0.085 | No | Non-ionic | V-I | Senofilcon-A | DMA, HEMA, PVP |
| 12 | Core33 /Surface80 |
140 | 156 | 14.1 | 8.7 | 0.09 | No | Non-ionic | V-IV | Delefilcon-A | CE-PDMS, DMA, TRIS |
| 13 | Core51, /Surface80 | 90 | 100 | 14.2 | 8.5 | 0.09 | No | Non-ionic | V-IV | Verofilcon A | mPDMS, GPDMS, NVP |
| - | Correlation Coefficients | Multivariate Analysis | ||||||
|---|---|---|---|---|---|---|---|---|
| Variables | R | (95% CI) | P Value | OR | P Value | |||
| Water Content (%) | -0.48 | (-0.60 – -0.33) | <0.001 | 0.1 | 0.007 | |||
| Oxygen Permeability (Dk) | -0.43 | (-0.56 – -0.27) | <0.001 | 0 | 0.03 | |||
| Oxygen Transmissibility (Dk/L) | -0.42 | (-0.55 – -0.26) | <0.001 | 329.8 | 0.042 | |||
| Diameter (mm) | 0.14 | (-0.03 – 0.31) | 0.109 | 0 | 0.704 | |||
| Base Curve (mm) | 0.16 | (-0.02 – 0.32) | 0.075 | >100 | 0.002 | |||
| Center Thickness | -0.33 | (-0.48 – -0.17) | <0.001 | >100 | 0.023 | |||
| SCL color (clear=0/color=1) | - | - | - | >100.0 | <0.001 | |||
| Surface charge (non-ionic=0/ionic=1) | - | - | - | 0 | <0.001 | |||
| FDA classification | - | - | - | >100.0 | 0.002 | |||
3.2. Degree of Pollen Particles Adherent to SCL
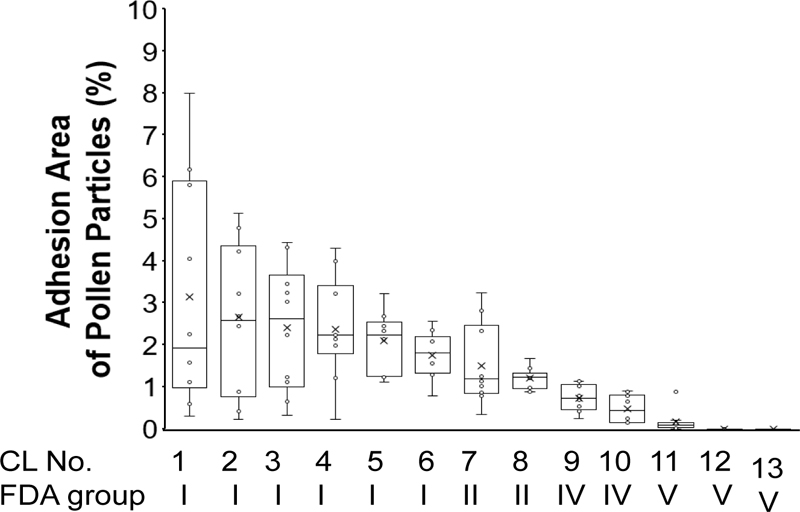
The percentage of the 200 × 200 um surface area of a SCL that was covered by pollen was 3.15 ± 2.55% in CL1, 2.65 ± 1.66% in CL2, 2.40 ± 1.43% in CL3, 2.38 ± 1.16% in CL4, 2.11 ± 0.66% in CL5, 1.76 ± 0.54% in CL6, 1.51 ± 0.90% in CL7, 1.21 ± 0.23% in CL8, 0.72 ± 0.29% in CL9, 0.48 ± 0.31% in CL10, and 0.16 ± 0.25% in CL11. The percentage of pollen adhered area was 3.16 ± 2.54% in CL1, 1.15 ± 1.11% in CL2, 3.07 ± 2.02% in CL3, 1.44 ± 1.24% in CL4, 3.15 ± 1.43% in CL5, 2.77 ± 2.16% in CL6, 0.61 ± 0.39% in CL7, 0.04 ± 0.07% in CL8, 0.51 ± 0.40% in CL9, 1.24 ± 0.77% in CL10, 0.66 ± 0.61% in CL11, 0.00 ± 0.00% in CL12, and 0.00 ± 0.00% in CL13 (Fig. 3). There was a significant difference among the 13 groups (one-way ANOVA, P<0.001).
3.3. Factors Affecting Adhesion of Pollen Particles to SCLs
There were significant negative correlations between the number of pollen particles adhered to the SCLs and some parameters of SCL, such as the water content (r = −0.48), oxygen permeability (r = −0.43), oxygen transmissibility (r = −0.42), and center thickness (r = −0.33) (Table 2).
Correlations between the number of pollen particles adhered to SCLs and parameters of SCLs were calculated with the two-tailed Pearson’s product moment formula. Independent determinants of the number of adhered pollen particles were investigated by multiple logistic regression analysis. R = Pearson’s correlation coefficient; CI = confidence interval; OR = odds ratio.
Fig. (4) shows the correlations between the portion of the pollen adhesion area and each characteristic of the SCLs. The area of pollen adhered to SCLs was significantly higher for colored SCLs than for clear SCLs (2.73 ± 1.97% vs. 1.03 ± 1.01%, P<0.001). The area of pollen adhered to SCLs was negatively correlated with water content (r = −0.51), oxygen permeability (r = −0.54), oxygen transmissibility (r = −0.54), and center thickness (r = −0.42).

The pollen adhesion area was smaller in the silicone hydrogel SCLs (SCL11–13; 0.05 ± 0.16%) than in the hydroxyethyl methacrylate HEMA SCLs (CL1 to CL10, 1.84 ± 1.45%, P<0.001). The portion of pollen adhesion area was lowest in both silicone hydrogel SCLs made with delefilcon-A (0.00 ± 0.00%) and verofilcon-A (0.00 ± 0.00%) (Fig. 4).
4. DISCUSSION
Our results showed that the number of pollen particles adhered to 13 different 1DSCLs varied from 0 to 185 particles per 200 × 200 µm area. Pollen adhesion was higher in the colored SCLs and lower in the silicone hydrogel SCLs. These results indicate that the degree of pollen adherence to the surface of the new verofilcon-A clear silicon hydrogel SCLs was very low.
In general, SCLs made of silicone hydrogel have the advantages of high oxygen permeability and resistance to drying [15]. However, some SCLs made of solid materials are still not comfortable to wear. To overcome the weaknesses of these silicone hydrogel materials, verofilcon-A SCL has succeeded in improving the wearing comfort with its unique technology called “SMARTSURFACE™ Technology” [16]. The surface of the verofilcon-A SCL has a high water content of 80% or more, which is accomplished by covering the surface of the SCL with a highly water-retaining hydrophilic polymer. SMARTSURFACE® is based on a manufacturing process in which the CLs are immersed in a liquid filled with a water-soluble polymer containing a hydrogel polymer and polyacrylic acid (PAA). PAA is a very hydrophilic polymer capable of absorbing large amounts of water, producing a “hydrogel.” PAA is a major contributor to the high water content of verofilcon-A SCL, which has a 51% water core wrapped in a gel-like outer layer that transitions to 80% at the surface [16]. In the processing stage of SMARTSURFACE™ Technology, this hydrophilic polymer solution expands the lens material to create small and narrow pores on the lens surface that are 2–3 microns in diameter [16]. The water-loving PAA polymer penetrates the open pores and is locked into place, creating a micro-thin layer of moisture. In addition, the heating process crosslinks the PAA polymer with a wetting agent consisting of a copolymer of polyamide amine and polyacrylamide-acrylic acid. This thin layer of PAA on the lens surface provides long-lasting moisture, support for stabilizing the tear film, and smoothness of the lens surface. These properties lead to long-lasting comfort and clear vision, according to the company’s literature [16]. The moderately hard structure in the center of the SCL makes it easier for SCL wearers to handle it when inserting and wearing these SCLs. In fact, 82% of new SCL wearers have reported that verofilcon-A SCLs are easy to place on the cornea and 72% agreed that they are easy to remove at the end of the day [17]. Furthermore, in a questionnaire survey of SCL wearers, verofilcon-A SCLs were reported to provide high quality of vision, comfort of wearing, and easy handling of wearing [18, 19]. SMARTSURFACE™ Technology is expected to not only improve the comfort of wearing SCLs but also reduce the adhesion of proteins and air dust on the SCL surface. In fact, our results showed that verofilcon-A SCLs had lower pollen adhesion than the other SCLs. As parameters for SCLs other than verofilcon-A, the water content, oxygen permeability, and oxygen transmissibility had a negative and significant correlation with the number of pollen particles adhered to the SCLs (Table 2). In addition, the colored SCLs and non-ionic HEMA SCLs had a high pollen adhesion number (Table 2). Generally, SCLs made with HEMA material have more protein adhesion than silicone hydrogel SCLs [20]. Because the silicone materials are hydrophobic and lipophilic, the silicone SCLs repel tears and are prone to the adherence of lipid particles. Therefore, to improve the wettability of the SCL surface, silicone hydrogel SCLs are coated with a hydrogel-rich material [20]. SCLs are coated with a variety of substances after being soaked in a dispersion of positively charged particles (DOWEX™) [20]. Therefore, positively charged pollen particles do not easily adhere to the SCLs treated with a positively charged material.
We used SCLs made of different materials: polymacon, omafilcon A, hilafilcon-B, etafilcon-A, ocufilcon-D, senofilcon-A, delefilcon-A, and verofilcon-A. All of these are very popular SCLs available in Japan. Of the 13 types of lenses, 11 lenses, excluding delefilcon-A and verofilcon-A, were different types of SCLs from the SCLs examined in earlier studies [12]. Nevertheless, the results regarding pollen adhesion to the SCL surface were similar to those of earlier studies using different lenses [12]. These results support the idea that SCL materials are involved in the degree of pollen adhesion to SCLs. That is, SCLs of non-ionic HEMA material, low water content, low oxygen permeability, and low oxygen transmissibility are those that pollen tends to adhere to.
The pollen adhesion area was not related to the base curve of the SCLs (Fig. 4). We examined only the center part of the SCL; therefore, the curvature of the entire surface of the SCLs probably did not affect pollen adhesion. In addition, there were only 4 types of lens curvatures, 8.5 mm, 8.6 mm, 8.7 mm, and 8.75 mm, which may have contributed to the absence of statistical significance. The center thickness of the SCLs was negatively correlated with the degree and area of pollen adhesion (Fig. 4). The center thickness of SCLs ranged from 0.05 to 0.09 mm (Table 1). The thickness of silicone hydrogel SCLs with less pollen adhesion was very thick (0.085–0.09 mm, CL11, CL12, and CL13; (Table 1). Therefore, the relationship between SCL center thickness and pollen adhesion may be influenced by the properties of the SCL material, especially the silicone hydrogel material.
This study has several limitations. First, the amount of pollen used was much higher (approximately 2,500 pollen/mm2 dropped on the surface of SCLs) than the general amount of pollen floating in the air (approximately 100 particles/cm2/day in Tokyo from March to April 2021). Second, the pollen adhesion experiments were only verified using 1DSCLs. The properties of 2-week and monthly replacement SCLs or conventional SCLs deteriorate over a long period of use and easily adsorb proteins. Therefore, rather than the 1DSCLs, a study of pollen adhesion to the 2-week frequent replacement SCLs or conventional SCLs may be useful for CL users.
CONCLUSION
In conclusion, the adherence of pollen and protein to SCL was lowest for verofilcon-A processed with SMART SURFACE™ Technology. Thus, verofilcon-A, like the delefilcon-A, maybe the best SCL for users with hay fever or allergic conjunctivitis.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
This study was reviewed and approved by the Ethics Committee of Teikyo University, Tokyo, Japan (#Teirin 20-166).
HUMAN AND ANIMAL RIGHTS
No animals were used in this research. All human research procedures were followed in accordance with the ethical standards of the committee responsible for human experimentation (institutional and national), and with the Helsinki Declaration of 1975, as revised in 2013.
CONSENT FOR PUBLICATION
Not applicable.
AVAILABILITY OF DATA AND MATERIALS
The data that support the findings of this study are available from the corresponding author [T.M], upon reasonable request.
FUNDING
This work was supported in part by a Grant-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology of Japan (grant number 20H04347) and an unrestricted investigator-initiated grant from Alcon Japan Ltd. to Tatsuya Mimura, MD.
CONFLICT OF INTEREST
Dr. Tatsuya Mimura is the Editor-in-Chief of The Open Ophthalmology Journal.
ACKNOWLEDGEMENTS
We would like to thank Editage (www.editage.com) for English language editing.


