All published articles of this journal are available on ScienceDirect.
Age-related Changes of the Corneal Endothelium in the Hispanic Elderly Population
Abstract
Purpose:
To assess the corneal endothelial morphometry in healthy elderly Hispanic eyes.
Methods:
A retrospective, cross-sectional, observational, descriptive, and analytical study was designed to assess the central region of the corneal endothelium by specular microscopy. A total of 241 eyes from 125 patients were studied. All eyes included were from healthy patients 65 or older (mean: 74.56± 6.74), without ocular disease or surgeries.
Results:
The mean cell density (MCD) was 2198.92±493.43 cells/mm2. 125 (51.87%) eyes had polymegethism, with a mean coefficient of variation (CV) of 42.89 ± 9.16%. 148 (61.41%) eyes presented pleomorphism, with an average of 46.25 ± 7.13% hexagonal cells. The mean corneal thickness was 527.55±28.37 μm. A statistically significant difference between age groups was found for MCD and pachymetry (P<0.05). 41 eyes (17.01%) had cornea guttata.
Conclusion:
This study suggests that the elderly Hispanic population has a high prevalence of polymegethism, pleomorphism, and guttata. Surprisingly, the oldest age group (>85) had an above-average MCD, with lower rates of polymegethism and pleomorphism.
Trial Registration Number: #ARCC/ECH2019
Date of Registration: October 18, 2019
1. INTRODUCTION
The corneal endothelium is an essential part of the cornea’s structure and function, playing a key role in maintaining corneal hydration homeostasis and transparency.
The measurement of central corneal thickness (CCT), endothelial cell density, size, and morphology is essential for evaluating the endothelial cell function for diagnostic purposes and the preoperative evaluation of any anterior segment surgery [1]. The total number of endothelial cells may be affected by aging, trauma, UV exposure, inflammation, surgery, etc [2-4].
Corneal endothelial cell alterations may affect corneal transparency, worsening visual outcomes. Age plays an essential role in this process since it has demonstrated a direct correlation with endothelial cell loss by reducing pump functionality and corneal wound-healing, which contribute to reduced visual acuity [3, 5].
Various studies analyzing the corneal endothelial cell density in different populations highlight the importance of including the endothelial cell morphology in relation to ethnicity and age. Available data from these studies show statistically significant differences in endothelial cell parameters among ethnic groups and sexes [1, 6-13].
The differences in endothelial cell morphology among ethnic groups, age, and sex show that normative data on the endothelial cell parameters may help assess the endothelial cell function in individual patients and further our understanding of corneal endothelial diseases.
The present study aims to describe the cell density and morphological characteristics of the corneal endothelium of the healthy elderly Hispanic population.
2. SUBJECTS AND METHODS
A retrospective, cross-sectional, observational, descriptive, and analytical study was designed to study the specular microscopy characteristics of the corneal endothelium in elderly Hispanic patients. The study was conducted at the Tecnologico de Monterrey Institute of Ophthalmology and Visual Sciences between 2013 and 2019. All patients enrolled in the study participated voluntarily after reading and signing a written informed consent, previously reviewed and approved by our institution’s Ethics and Research Committees (#ARCC/ECH2019), following the tenets of the Declaration of Helsinki.
The inclusion criteria comprised Hispanic patients over 65 years of age, living in Northern Mexico who came for a routine ophthalmological examination and were otherwise healthy. The exclusion criteria included a past medical history of ocular trauma, corneal or intraocular inflammation, the use of systemic drugs that could affect endothelial cells, evidence of endothelial disease during a slit-lamp examination, previous ocular surgery (including cataract surgery), increased intraocular pressure, corneal opacity, or retinal photocoagulation procedures.
After a complete medical history and general ophthalmological examination, all eyes of patients included in the study had specular microscopy (SM) performed by the same technician (SIS) using the EM-3000 specular microscope (Tomey®, Phoenix, AZ, USA) at a similar room temperature and humidity conditions. EM-3000 is a non-contact specular microscope that uses a charge-coupled device camera to capture 15 images in a series; it automatically selects and displays the finest among these images. The in-built software allows for fast and precise analysis of endothelial parameters. A wide range capturing area of 0.25 x 0.54 mm allows a larger count of cells. Corneal thickness measurement accuracy is +/- 10 μm.
Only the central corneal measurement point was considered for analysis. Parameters studied included the mean endothelial cell density (MCD), the mean cell area (MCA), the coefficient of variation (CV) of cell area (calculated as SD/mean cell area in μm), and the percentage of hexagonal cells (PHC), and the CCT. Polymegethism was considered a CV over 40% and pleomorphism when less than 50% of the endothelial cells showed hexagonality. SM images were classified as healthy when minor dark spots were present or as having guttae, mound-shaped excrescences in the Descemet's membrane.
Patients were classified according to age into five groups: 65-69, 70-74, 75-79, 80-84, and ≥85 years old. All statistical analyses were performed using STATA v. 12.0 (StataCorp® LLC, Texas, USA). Continuous data are presented in mean ± SD and categorical data as proportions. Since the data showed an abnormal distribution according to the Shapiro-Wilk test and distribution plots, Mann-Whitney U-test was performed to compare cell parameters according to gender and Kruskal-Wallis one-way analysis of variance to compare morphometry parameters between age groups. A multiple regression analysis was conducted to determine endothelial cell density changes with age, CV, MCA, PHC, and CCT. A P value less than 0.05 was considered statistically significant.
3. RESULTS
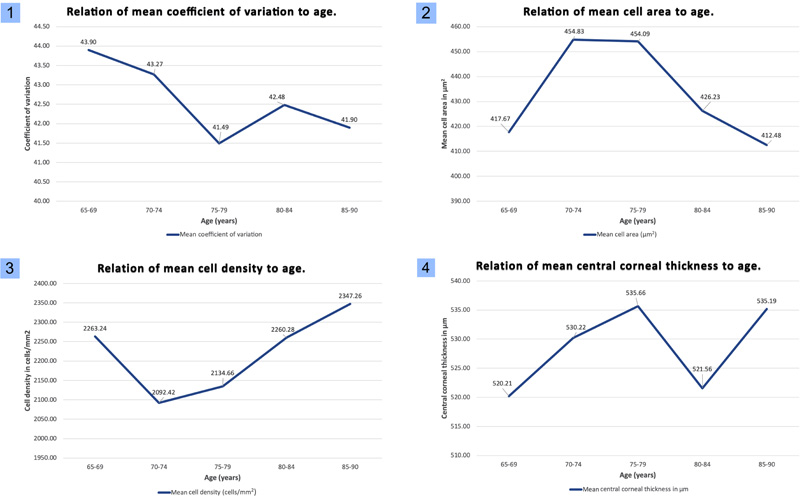
A total of 241 eyes from 125 patients corresponding to 64 (51.20%) males and 61 (48.80%) females over 65 years old, were studied. Nine pairs of eyes from nine patients had undergone previous cataract surgery and were therefore excluded for statistical analysis. A mean number of 219.46± 66.1 endothelial cells were counted. The endothelial cell density and morphology according to patients' age distribution are shown in Table 1. Fig. (1) shows a collage of morphometrical parameters in relation to age.
| Age group (y) | No. of eyes | Mean age (y) | MCD (cells/mm2) | MCA (μm2) | MCV (%) | Hexagonality (%) | MCCT(μm) | Mean no. of cells |
|---|---|---|---|---|---|---|---|---|
| 65-69 | 63 | 66.8±1.5 | 2263.2±509.9 | 417.6±110.8 | 43.9±9.1 | 45.5±7.5 | 520.2±25.3 | 226.7 |
| 70-74 | 77 | 71.7+1.4 | 2092.4±487.8 | 454.8±157.3 | 43.2±10 | 46.3±7.3 | 530.2±26.1 | 209.4 |
| 75-79 | 35 | 77.4+1.5 | 2134.6±466.4 | 454.1±166.3 | 41.5±5.9 | 44.6±6.5 | 535.6±34 | 216.3 |
| 80-84 | 39 | 82.1±1.6 | 2260.2± 522.9 | 426.2±154.5 | 42.5±10.4 | 47.1±7.7 | 521.5±30.8 | 234.3 |
| 85-90 | 27 | 85.9±1.1 | 2347.2±412.1 | 412.4±88.7 | 41.9±8.3 | 48.5±4.9 | 535.1±25.6 | 210.3 |
3.1. Mean Cell Density
The MCD across all age groups was 2198.92±493.43 cells/mm2 (95% CI 2137 to 2261). A small decline was observed as age increased, except for the ≥85 years group, where surprisingly, an increase in cell density was observed (MCD: 2347.2 ± 412.1). The changes in MCD among age groups were statistically significant (P=0.003). MCD was higher in males than in females (2275.7 vs. 2120.1 cells/mm2, P= 0.02). Fifteen eyes (6.22%) had a CD over 2800 cells/mm2, while thirty (12.25%) had less than 1500 cells/mm2; of those, only five (2.07%) had fewer than 1000 cells/mm2.
3.2. Mean Cell Area
The MCA across all age groups was 435.63 ± 141μm2 (95% CI 417.8 to 453.4). No statistically significant difference in MCA was observed among age groups (P=0.24).
3.3. Mean Coefficient of Variation
The MCV for all age groups was 42.89 ± 9.16% (95% CI 41.73 to 44.05). No statistical difference was observed among groups for MCV (P=0.73). The 65-69 years age group had the highest prevalence of polymegethism, while the 80-84 years age group had the lowest (60.32% vs. 38.4%, respectively).
3.4. Mean Hexagonality
The PHC was 46.25 ± 7.13% (95% CI 45.35 to 47.15). No statistical difference was observed among groups for PHC (P= 0.09). The 75-79 years age group had the lowest mean PHC, while the ≥85 years group had the highest (44.63% vs. 48.48%, respectively). Age was not a risk factor for pleomorphism (R=0.005, p=0.27) in this population. The 75-79 years age group had the highest prevalence of pleomorphism (77.14%), while the ≥85 years age group had the lowest (44.4%).
3.5. Mean Central Corneal Thickness
The mean CCT for all age groups was 527.55 ± 28.37μm (95% CI 524 to 531.1). A steady increase in CCT was observed in relation to age (R=0.012, P=0.09). A statistically significant difference was observed among age groups for the mean CCT (P=0.02). The lowest mean CCT value was recognized in the 65-69 years age group (520.2 ± 25.3μm) and the highest in the 75-79 years age group (535.6 ± 34μm)
Finally, a total of 41 eyes (17%) showed cornea guttata. A multiple regression analysis was run to predict cell density based on age, CV, MCA, PHC, and CCT: F (5,69) = 154.29, p= <0.0001, R2= 0.9179. Variables, CV, MCA, and PHC added statistical significance to the prediction (p=<0.05).
4. DISCUSSION
Corneal endothelial cells are essential for maintaining corneal transparency by keeping the corneal stroma in a state of partial dehydration. Such homeostatic status is accomplished by a delicate balance between the rate of water entering the hydrophilic stroma through the endothelial barrier and its active removal by the action of the metabolic Na/K ATPase endothelial pump [14].
| Parameter | Female | Male | P-value |
|---|---|---|---|
| Mean age (y) | 74.43±6.39 | 74.68±7.09 | 0.90 |
| MCD (cells/mm2) | 2120.1±519.7 | 2275.75±455.4 | 0.02 |
| MCA (μm2) | 454±159.60 | 417.69±118.1 | 0.24 |
| MCV (%) | 43.91±10.21 | 41.9±7.92 | 0.08 |
| Hexagonality (%) | 45.21±7.92 | 47.2±6.14 | 0.08 |
| MCCT(μm) | 529±29.68 | 526.1±27.1 | 0.41 |
| Population | Author | MCD (cells/mm2) | Age (y) | No. of eyes |
|---|---|---|---|---|
| Lithuania | Galgauskas1 | 2366 | 60 – 89 | 158 |
| Philippines | Padilla6 | 2780.45 | 61 – 86 | 200 |
| India | Rao9 | 2404 | 60 – 87 | 142 |
| Turkey | Goktas10 | 2,215 | 61 – 90 | 371 |
| Iran | Hashemian11 | 1681.61 | 61 – 85 | 225 |
| Central Mexico | Graue12 | 1805.33 | 60 – 89 | 25 |
| Central Mexico | Molina13 | 1910 | 60 – 89 | 50 |
| North Mexico | Valdez et al. | 2198 | 65 – 90 | 241 |
After birth, the corneal endothelial cell density (ECD) peaks at approximately 6000 cells/mm2 which starts to decline during the first years of life due to corneal growth [15]. Then, cell density continues its gradual decay over time, particularly between the ages of 20 to 80 years at an estimated annual rate of 0.6% [5]. This slowly progressive cell loss is accompanied by a proportional increase in polymorphism and polymegethism [16].
When aging, toxicity, trauma, inflammation, or disease produce an ECD drop below 500 cells/mm2, irreversible stromal edema occurs, and the cornea eventually loses transparency [17]. For many years it was thought that corneal endothelial cells could not regenerate. However, recent investigations have shown that endothelial cells retain their capacity to divide and renew, although they rarely do so [18]. A hypothesis that may help explain, at least in part, such cellular behavior postulates that certain groups of dividing cells may enter a process of cellular senescence or replicative failure that limits their capacity to maintain their lifespan [19, 20]. This hypothesis is supported by an age-related increase in the number of senescent cells in the human corneal endothelium [21, 22]. Despite the relentless pursuit of a better understanding of the biologic mechanisms implicated in the gradual endothelial cell loss found during aging, there are still more questions than answers. This phenomenon is probably multifactorial, involving environmental, hormonal, biochemical, and physiological processes related to aging [23].
Several studies from diverse populations worldwide analyze the corneal endothelial cell density and morphology. Although investigators differ in their findings concerning the relationship between age and gender and the corneal endothelial characteristics, the literature clearly shows a significant difference in corneal endothelial properties among races and ethnic groups [6-9].
In this study, the MCD of healthy senior Hispanic patients was found to be within normal values for healthy corneas [24]. Our mean number of cells studied per patient is higher than reported by other authors.
The MCD among various elderly populations from other countries is shown in Table 3. Although none of the authors studied exclusively elderly patients, a stratified calculation based on their age groups was performed. The MCD of 2198.92 cells/mm2 found in our study across all age groups is similar to the Caucasian population in Lithuania (2366 cells/mm2) [1], the Turkish population studied by Goktas et al. [10] (2215 cells/mm2), and distant from the findings of Hashemian et al. [11] in Iran (1681.61 cells/mm2), and also from Padilla et al. in the Philippines (2780.45cells/mm2) [6]. .
It is important to mention the results of Graue et al. [12] and Molina et al. [13] in the Mexican population from Central Mexico, where patients in the 60-89 years age group showed an MCD of 1805.33 cells/mm2 and 1910.42 cells/mm2, respectively, contrasting with our findings and suggesting that corneal endothelial cells characteristics may differ with our counterparts in the central and southern region of the country. Another study in a population from Central Mexico showed an MCD of 1909 cells/mm2 in patients with a mean age of 71 years; interestingly, this cohort of patients had unilateral pseudophakic bullous keratopathy that developed after cataract surgery [25]. These geographical differences in MCD among the Mexican population may be clinically significant. However, further studies are needed to fully understand the mechanisms behind this disparity.
No decrease in MCD was found in our population as patients got older, suggesting that Hispanic patients who maintain ocular health after 65 years can also preserve a fair number of endothelial cells. The latter is supported by the fact that 73% of patients had a cell density of over 2000 cells/mm2, and only 12.45% showed an MCD <1500 cells/mm2. It has been shown in previous reports that as the mean population's age studied increases, there is also an increased spread in the range of MCD counts, making the endothelial cell density measurement an unreliable index for the evaluation of corneal aging. The same finding applies to other animal species studied, including dogs, cats, monkeys, and rabbits, where the density and morphology of endothelial cells change with age, but the adult MCD remains constant [22, 23, 26].
As for the endothelial cell size and shape, our population showed a high degree of polymegethism and pleomorphism (MCV of 42.89% and PHC 46.25%). These findings correlate with the fact that in most populations studied, pleomorphism and polymegethism tend to increase as patients age [27-30] and suggest that elderly Hispanic patients could have higher rates of pleomorphism and polymegethism than other ethnicities [1, 6-13]. Surprisingly, the group of ≥85 years showed a fair rate of polymegethism and pleomorphism (51.85% and 44.4%, respectively).
Regarding CCT 527.55 (95% CI 524 to 531), we find it interesting that these values are similar to those reported in a previous study performed by our group in younger patients (mean age: 32.54 ± 12.04 years and Mean CCT: 545.69 ± 36.88 μm). The findings reported in both studies support the idea that the Mexican population could present a different mean CCT, and “normal” CCT could be redefined for clinical purposes in our context [31].
Further, 17% of eyes had cornea guttata. An exploratory analysis of these eyes revealed an MCD of 1611.63 cells/mm2 and a mean CCT of 537.75, suggesting that cornea guttata was only a finding in these asymptomatic patients, and guttae might be associated to lower MCD values.
Some authors have found differences between gender in endothelial cell parameters [6-9]. As resumed in Table 2, our results show that those differences are only statistically significant for MCD.
Limitations of this study are the small but otherwise healthy senior population, and the study’s retrospective nature. Another further consideration is the possible presence of selection bias since specular microscopy is not performed as a routine examination in our country, and the cost may be prohibitively expensive to some patients.
CONCLUSION
However, we believe further prospective studies with control groups may be beneficial within the same population. This may be eased as specular microscopy becomes more available for the Mexican population.
In conclusion, the endothelial cell characteristics of elderly Mexican Northeastern patients differ from their counterparts from central and south Mexico and other nations. A high prevalence of polymegethism, pleomorphism, and guttata is present, but not in the >85-year age group, where a lower rate of these characteristics was described within the sample size studied.
AUTHORS’ CONTRIBUTIONS
All authors fulfilled ICJME criteria of authorship.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The study was approved by the Ethics and Research Committees (#ARCC/ECH2019).
HUMAN AND ANIMAL RIGHTS
The study was conducted following the Declaration of Helsinki principles.
CONSENT FOR PUBLICATION
All patients enrolled in the study participated voluntarily after reading and signing a previously written informed consent.
STANDARD OF REPORTING
STROBE guidelines have been followed in this study.
AVAILABILITY OF DATA AND MATERIALS
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
FUNDING
This research project received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
CONFLICT OF INTEREST
The authors declare no competing interests.
ACKNOWLEDGEMENTS
None.


