All published articles of this journal are available on ScienceDirect.
Anatomical Basis of Clinical Manifestations Seen in Cavernous Sinus Syndrome: A Narrative Review
Abstract
Pathology involving the Cavernous Sinus (CS), generally referred to as Cavernous Sinus Syndrome (CSS), can arise from vascular disorders, neoplasms, infections, and non-infectious inflammatory disorders. An acute understanding of the CS and its regional anatomical structures is therefore pertinent in expounding the highly variable clinical manifestations seen in CSS as well as laying the groundwork for surgical intervention. Though the neuroanatomy of the cavernous sinus has been substantially chronicled in literature, their correlation to clinical signs has only been minimally described. This narrative review serves to address this knowledge gap and aims to comprehensively correlate the clinical manifestations of CSS with the relevant neuroanatomy, thereby allowing medical practitioners to better navigate the diagnostic quandary. We conducted a scoping review of the literature concerning CS anatomy and CSS, complied through MEDLINE/OVID and cross-referencing of articles on PubMed and Google Scholar with the keywords cavernous sinus, cavernous sinus syndrome, clinical signs/manifestations, neuroanatomy, CS pathology, Cavernous Sinus Thrombus (CST) and cavernous sinus neoplasms/tumours.
1. INTRODUCTION
Cavernous sinuses are small, paired venous structures, each measuring approximately 3 cm long, 1 cm wide, and 0.5 cm high, that lie on either side of the pituitary fossa. These structures hold significance due to their close regional associations with various structures such as the internal carotid arteries, pituitary gland and cranial nerves [1-3]. Often in medical literature, clinical manifestations of diseases affecting the cavernous sinus and its closely related structures are broadly termed as the Cavernous Sinus Syndrome (CSS). Aetiologies of CSS can be further categorized as vascular disorders, neoplasms, infections, and non-infectious inflam- matory disorders [3, 4]. Though the neuroanatomy of the cavernous sinus has been substantially chronicled in the literature, there is minimal literature to describe their correlation with clinical manifestations. This was our motiva- tion to carry out a scoping review to explore and illustrate the normal anatomy of the cavernous sinus and comprehensively correlate the clinical features of CSS with the relevant neuroanatomy.
2. MATERIALS AND METHODS
A scoping review was carried out on PubMed, Ovid Medline, and Goggle Scholar till December 2019 by PRR and vs. Inclusion criteria for articles were primarily on those focusing on CS neuroanatomy, CSS, and the associated clinical manifestations. Keywords utilised included “cavernous sinus syndrome”, “cavernous sinus”, “pathology”, “neuroanatomy”, “ophthalmoplegia”, “clinical signs”, “cavernous sinus thrombus”, “cavernous sinus neoplasms” and “clinical manifestations”. Our focus was on both primary research articles and review articles pertaining to human subjects. No limits were placed on the language used. Reference lists were also reviewed to source additional data. Illustrations are all original content.
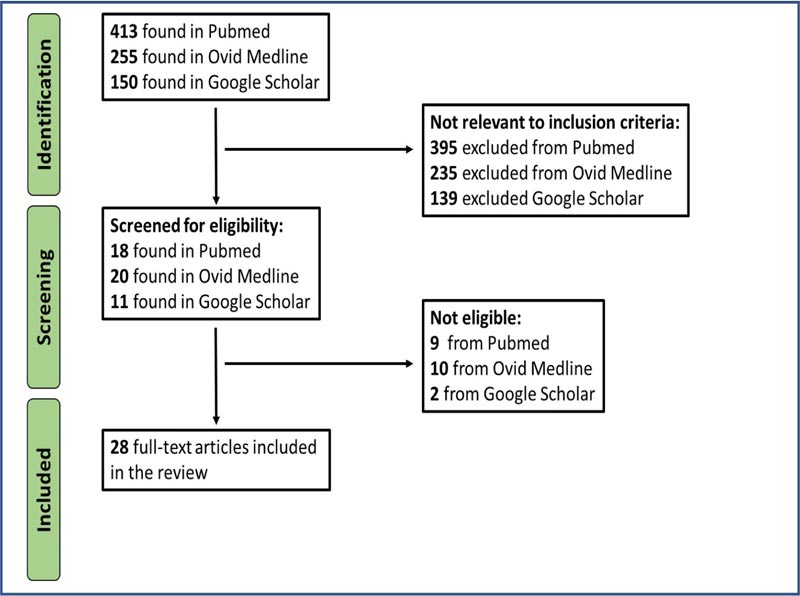
A total of 818 citations were reviewed. Articles to be included in the review were selected in consensus between PRR and vs. 28 articles fulfilled the inclusion criteria for the review. The search strategy is documented in Fig. (1).
3. DISCUSSION
3.1. Normal Anatomy of the Cavernous Sinus
Foetal development of the CS commences during the stage of 70-95 mm crown-rump (13-15 weeks’ gestation) where the CS is noted to be a cluster of miniature non-uniform venous channels scattered in the immature mesenchyme of the developing lateral sellar compartment. These primitive venous channels, which include the primitive tentorial sinus, prootic sinus and the primitive superficial middle cerebral vein (SMCV), continue to enlarge and proliferate and their subsequent coalescence over 25-28 weeks’ gestation gives rise to the mature CS, a system of interconnecting trabeculated venous caverns around the internal carotid artery (ICA) that are all encapsulated by a dural fold [5, 6].
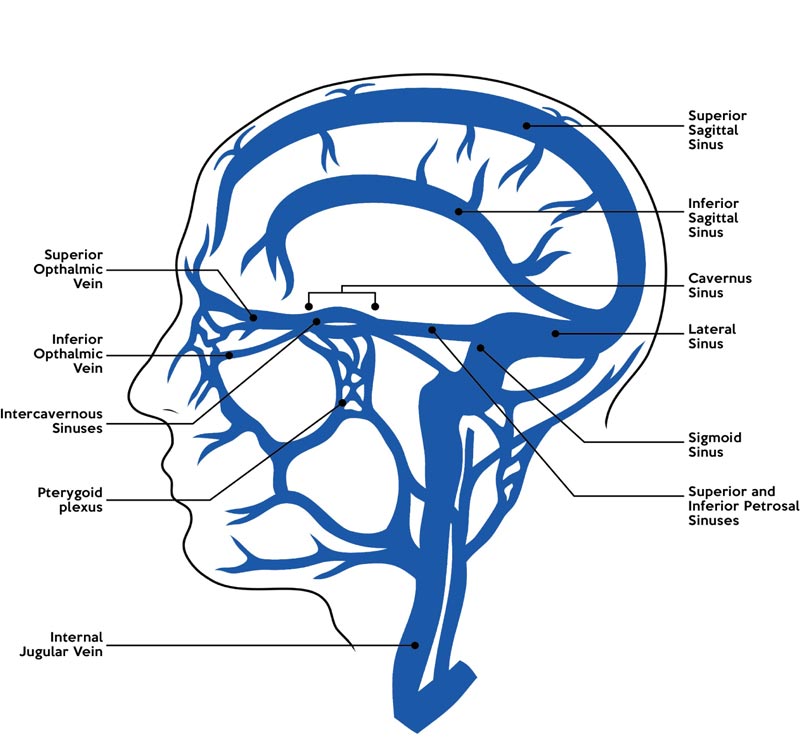
As shown in Fig. (2), the CS has communications with multiple extra-cavernous venous channels, with vascular input from the Superficial Middle Cerebral Vein (SMCV), the Sphenoparietal sinus (SPS), the superior ophthalmic vein and emissary veins from the pterygoid plexus [3, 5, 7]. The vascular output from the CS voids into the superior and inferior petrosal sinuses posterolaterally, the extracranial pterygoid plexus anteriorly and basilar plexuses posteromedially. The intercavernous sinuses situated behind and in front of the pituitary stalk connect both the left and right CS [3, 5, 7]. Several anatomical variants involving the input connections of the SMCV and SPS to the CS have been described in the literature and are attributed to the developmental anomalies that occur during embryological anastomosis of primitive venous structures [5]. Knowledge of these variants holds significance as they help guide surgical management of para-cavernous lesions and arteriovenous (AV) fistulas, where they offer potential reflux channels [5, 6].
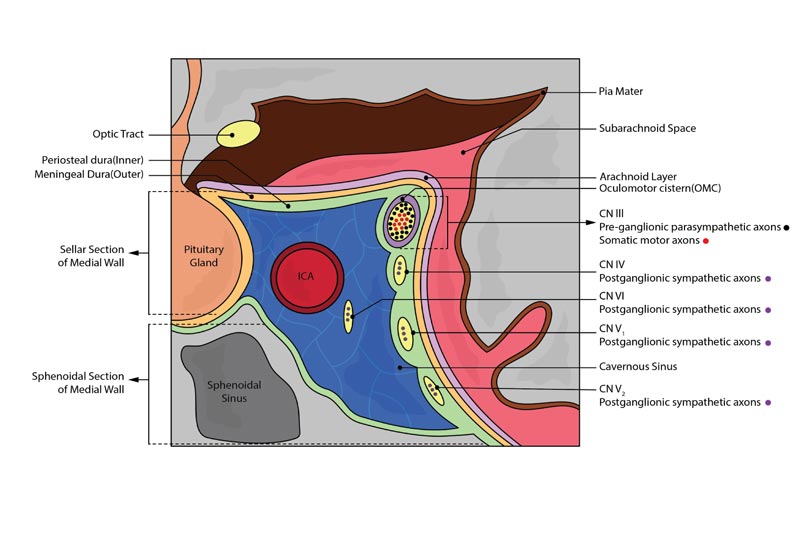
The external structure of each CS consists of four walls lined with dura – lateral, medial, superior, and posterior. As depicted in the coronal section of the CS in Fig. (3), the lateral wall of the CS is lined with two dural layers – the meningeal dura (outer) and periosteal dura (inner) [8, 9]. The inner layer of the lateral wall, which can be surgically divided from the outer layer, houses the oculomotor nerve (CN III) most superiorly, followed by the trochlear nerve (CN IV), ophthalmic division of the trigeminal nerve (CN V1) and the maxillary division of the trigeminal nerve (CN V2). These cranial nerves course through the superior orbital fissure (SOF) except CN V2, which exits via the foramen rotundum to eventually exit the CS. CN V1 has 3 branches (Frontal nerve, lacrimal nerve, and nasociliary nerve) at the anterior end of the sinus prior to its entry into the SOF [8-10].
As shown in Fig. (3), the medial wall of the CS can be divided into two parts – the sellar and sphenoidal sections. As opposed to the other walls, the medial wall is only lined with one dural layer. The sellar portion of the medial wall serves as a safe limiting landmark when surgically accessing the tunica sellar via a trans-sphenoidal approach. The sellar portion of the medial wall with its single dural layer is also anatomically a “weak spot” where it is vulnerable to lateral expansion of pituitary tumors into the CS [8].
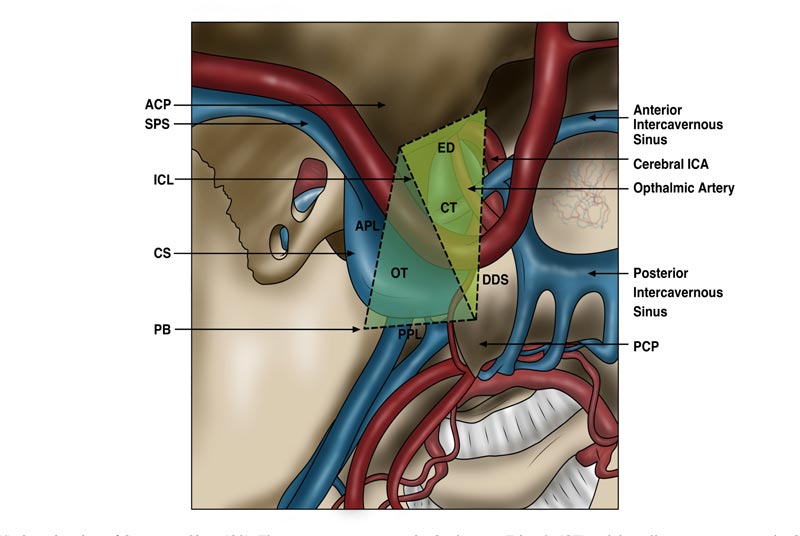
The roof (superior wall) of the CS is shaped like a trapezium and, like the lateral wall, it has dual dural layers. Its boundaries are formed laterally by the anterior petroclinoid ligament, medially by the lateral aspect of the diaphragm sellar, anteriorly by the inferior aspect of the anterior clinoid process and posteriorly by the posterior petroclinoid ligament [8, 9]. As shown in Fig. (4), it can be divided further into 2 triangles – the carotid triangle (CT) anteromedially and the oculomotor triangle (OT) posterolaterally. It is through the latter by which CN III and IV enter the CS [9].
Unlike the CN IV, CN III enters the roof of the CS while being encapsulated by the oculomotor cistern (OMC) at an opening termed the “porus” and remains within this cistern through the entirety of its course within the superolateral wall of the CS [11]. The trochlear cistern runs inferior to the OMC and terminates prior to CN IV’s entry into the CS [12]. The OMC is of clinical significance as it enables an avascular and safe approach to the surgical dissection and mobilisation of CN III within the cavernous sinus [11]. CN III also carries parasympathetic pre-ganglionic nerve fibres that upon exiting the SOF, synapse in the ciliary ganglion situated in the posterior orbit [10].
The posterior wall of the CS, like the lateral and superior walls, is made up of two dural layers. It runs anteriorly and continuously with the posterior aspect of the OT and is related medially to the basilar sinus and laterally with the dura mater covering the petrous bone [8]. CN V1 and V2 pierce the posterior wall inferiorly and continue toward the SOF in the lateral wall of the CS [8, 10]. The abducens nerve (CN VI) also enters the CS through the posterior wall inferiorly after leaving Dorello’s Canal. Unlike the other cranial nerves that course within the lateral wall of the CS, CN VI courses centrally through the CS inferolateral to the internal carotid artery (ICA). Being centrally located within the CS, CN VI is particularly vulnerable to intracavernous pathology such as cavernous ICA (C4 segment) aneurysms [13, 14].
As depicted in Fig. (5), the ICA enters the inferior aspect of the CS at the level of the petrolingual ligament, accompanied by the sympathetic plexus originating from the superior cervical sympathetic ganglion. Within the CS, the ICA can be divided into the posterior vertical segment, posterior genu, horizontal segment, anterior genu and anterior vertical segment [14, 15]. Whilst in the CS, postganglionic fibres from the sympathetic plexus are passed on to CN III, CN IV, CN V1, CN V2 and CN VI and eventually enter the orbit via the SOF. A large proportion of postganglionic sympathetic fibres also enter the orbit via the ophthalmic artery, which normally branches off the C6 segment of the ICA after the ICA exits the CS superiorly [10, 14].
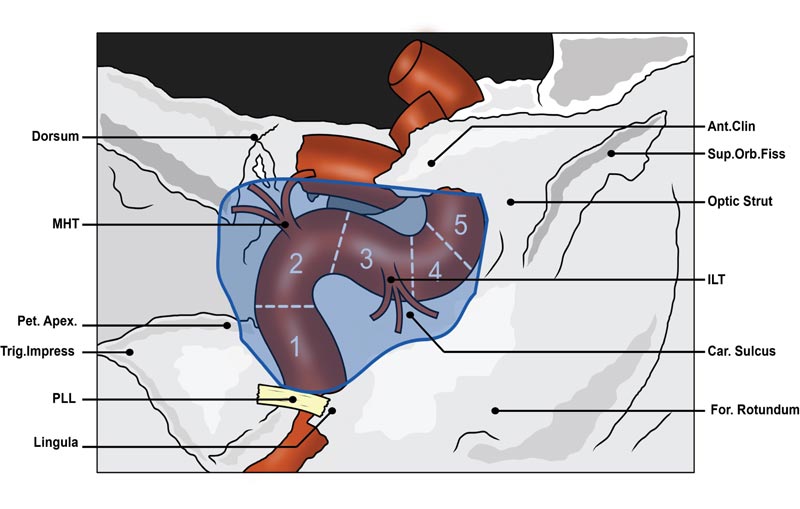
Traditionally, the cavernous ICA has two main branches, the meningohypophseal trunk (MHT) and the inferolateral trunk (ILT), which originate from the posterior genu and the lateral aspect of the horizontal segment of the ICA, respectively. Considering anatomical variants, the MHT and ILT generally give off 3-4 branches. These branches supply the intracavernous cranial nerves and dura as well as contribute vascular supply to extracavernous structures such as the pituitary gland and tentorium [1, 14].
3.2. Cavernous Sinus Syndrome (CSS)
Depending on the aetiology, CSS can present in a spectrum of clinical features, including ocular pain, external ophthalmoplegia (impairment of extra-ocular muscles), internal ophthalmoplegia (impairment of the iris and ciliary apparatus), diplopia, proptosis, Horner’s Syndrome, and trigeminal sensory loss in the distribution of CN V1 and V2 [1-4].
External ophthalmoplegia is a result of cavernous lesions affecting CN III, CN IV, or CN VI. A CN III palsy can cause impaired adduction, elevation, and depression of the eye due to paralysis of the medial rectus, superior rectus and inferior rectus, respectively [2, 16]. The impairment of these ocular muscles can also result in multi-directional diplopia [16]. An isolated CN III palsy classically results in the “down and out” eye due to the unopposed actions of the superior oblique and lateral rectus muscles that are supplied by CN IV and CN VI, respectively. A CN III palsy can also result in ptosis of the affected eye due to impaired functioning of the Levator Palpebrae Superiorus (LPS), which serves to elevate the eyelid [16, 17].
Impairment of preganglionic parasympathetic fibres that run with CN III can also result in pupil asymmetry due to paresis of the iris and the ciliary apparatus, with the affected eye appearing dilated and poorly reactive to light. However, in cavernous sinus lesions, pupil appearance varies greatly and may even appear normal due to concomitant oculosympathetic impairment given the proximity of the cavernous ICA sympathetic plexus and branching sympathetic fibres to intracavernous cranial nerves in the lateral wall of the CS [4, 10, 16].
Isolated CN IV and CN VI palsies mainly result in vertical and horizontal diplopia, respectively. In addition, esotropia is often seen in isolated CN VI lesions due to the unopposed action of the medial rectus muscle [16, 17]. CN V1 palsy results in the loss of sensation to the forehead and anterior scalp as well as loss of the corneal reflex. CN V2 palsy mainly results in the loss of cutaneous sensation to the lower eyelid, cheek, and upper lip [17]. Occasionally, severe neuralgic pain can also occur in the said distributions of V1 and V2 [18]. Given the relative proximity of the cavernous cranial nerves, particularly with CN III, IV, V1 and V2 in the lateral wall of the CS, isolated CN palsies are uncommon in CS lesions and patients often present with a combination of symptoms involving multiple intracavernous cranial nerves.
Horner’s Syndrome refers to the classic triad of ptosis, anisocoria (due to miosis of affected eye) and anhidrosis, all of which occur secondary to disruption of sympathetic pathways [19]. Post-ganglionic sympathetic fibres accompany the ICA into the cavernous sinus enroute to innervating the iris dilator and the superior and inferior tarsal muscles within the upper and lower eyelids, respectively. As such, in CSS, a disruption to the cavernous sympathetic pathway compromises the function of the iris dilator resulting in dilator lag and miosis of the affected eye (internal ophthalmoplegia) [10, 16, 19]. Also, consequent denervation of the superior tarsus muscle results in moderate ptosis due to impairment of the superior tarsus muscle. However, the degree of ptosis is not as profound as that observed in CN III palsies due to the preserved function of the LPS [16, 19]. The denervation of the inferior tarsus muscle can also result in an “up-side down” ptosis, which further narrows the palpebral fissure. Anhidrosis, however, is usually not seen in cavernous sinus lesions as sympathetic branches responsible for facial sweating and vasodilatation branch off separately at the superior cervical ganglion and do not contribute postganglionic fibres to the cavernous sympathetic pathway [19].
3.3. Cavernous Sinus Neoplasms
As outlined in Fig. (5), cavernous sinus (CS) neoplasms can be categorized based on their origin as (i) primary lesions within the CS, (ii) invading lesions from adjacent structures, and (iii) systemic metastases from extracavernous malig- nancies. CS neoplasm is the most common cause of CSS, with a majority, approximately 41%, being benign primary meni- ngiomas [20, 21]. In the paediatric population, neurinomas of CN V make up the clear majority of CS tumors [1, 18]. The direct invasion of tumors into the CS from surrounding areas such as pituitary adenomas, craniopharyngioma, nasopha- ryngeal carcinomas and sarcomas as well as perineural and hematogenous metastases from extracavernous malignancies such as lymphoma, multiple myeloma and histiocytosis are also notably known to cause CSS [1, 4, 18, 20].
Clinical features of CS tumors commonly include head- ache, palsies of cavernous cranial nerves including external and internal ophthalmoplegia, ptosis, diplopia, and neuralgic pain/numbness over V1 or V2. Headache and diplopia tend to occur early during tumor growth, with ophthalmoplegia seen in the late stages [18]. CN V2 impairment is considered very specific for cavernous sinus tumors and is postulated to be secondary to tumor invasion into the floor of the cavernous sinus inferolaterally (Fig. 1) [20]. Tumor type can also dictate clinical presentation, as seen with large pituitary adenomas that often cause endocrine symptoms such as hypogonadism, acromegaly, or galactorrhoea in conjunction with features of CSS.
Treatment of CS neoplasms is often guided by tumor origin, type, and function. Primary CS neoplasms such as meningiomas represent a high-risk surgical entity due to poor accessibility and frequent involvement of the cavernous ICA. Given the indolent nature of these tumors, regular surveillance without specific treatment is often the recommended approach in elderly patients and in patients with minor symptomatology [22, 23]. Partial resection of meningiomas is only considered in patients with debilitating symptoms such as intractable pain or complete ophthalmoplegia in patients with monocular vision [1, 23]. Gamma knife radiosurgery can also be used as an adjunctive treatment and has been shown to improve cranial nerve function [22, 23].
Invading neoplastic lesions from adjacent structures such as nasopharyngeal carcinomas and craniopharyngiomas often respond well to radiotherapy when performed in combination with partial/complete surgical resections, with prolonged periods of remission post-treatment [23]. Pituitary tumors encroaching the cavernous sinus can be surgically removed using an endoscopic transnasal-transsphenoidal approach or a transcranial approach, with the latter being a more invasive procedure reserved for larger complicated tumors [15, 22]. Adjunctive treatment with gamma knife radiosurgery can also be used for managing postoperative tumor residuals or tumor recurrence [22, 23]. Prolactinomas may improve with oral dopamine agonists such as cabergoline and bromocriptine without the need for immediate surgical intervention [23, 24]. Medications targeting cortisol production or function, such as mifepristone and pasireotide, can be administered to treat ACTH-secreting pituitary tumors where surgery is contra- indicated or unsuccessful [24]. Metastatic lesions from extracavernous malignancies are notoriously hard to eradicate, but targeted radiotherapy in conjunction with chemotherapy tailored to underlying primary malignancy may provide transient symptom relief [23, 24].
3.4. Cavernous Sinus Thrombosis/Thrombophlebitis (CST)
CST primarily results from the spread of bacterial or fungal infections in the face, oral cavity, paranasal sinuses, and orbits via superficial venous channels such as the facial veins and pterygoid plexus, which drain into the CS via the superior ophthalmic vein and emissary's veins, respectively. The pterygoid plexus also relates closely with deep facial muscles via the deep facial veins and the inferior ophthalmic vein [2, 25].
As shown in Fig. (2), the extensive input and output venous channels of the CS which include the dural sinuses and cerebral veins, are non-valvular in nature. As such, the direction of blood flow within the CS venous system is highly susceptible to change in pressure gradients due to infective thrombi, potentially allowing for extensive spread of infections from and into the CS [1-3, 22]. It has also been postulated that the trabeculated set-up of sinuses within the CS aids in trapping septic emboli and bacteria from the anteriorly infected sites [21].
CST is potentially a lethal cause of CSS and clinical manifestations can be either rapid or sub-acute. Initial clinical presentation often involves fever, malaise accompanied by unilateral chemosis, palpebral swelling and peri-orbital oedema, all of which are signs stemming from concomitant venous congestion secondary to infective inflammation within the CS and related sinuses and tributaries [2, 3, 25]. Ensuing pressure elevations in the CS and retrobulbar compartment result in further disease progression with the development of cranial nerve palsies, proptosis, internal ophthalmoplegia secondary to sympathetic/parasympathetic denervation and visual acuity changes due to retinopathy from central retinal vein congestion [2, 4, 25]. The contralateral eye may also exhibit features of CSS due to infection spread via the inter-cavernous sinuses. In addition, the progression of septic thrombosis to surrounding cerebral dural venous sinuses potentiates the development of elevated intracranial pressures and stroke symptoms [25].
The most implicated pathogen in CST appears to be Staphylococcus aureus, identified in approximately 60%-70% of patients. Though uncommon, streptococcal species, including Streptococcus pneumoniae, gram-negative bacilli and anaerobes, have also been implicated in CST [22, 25]. Positive blood cultures are evident in over 70% of cases, particularly in patients with acutely severe disease. Cerebrospinal fluid samples are only culture positive in about 20% of cases, though a vast majority appear abnormal with raised white cell counts and protein levels [25]. Very rarely, fungal species such as Aspergillus and Mucoraceae have been implicated as causative agents in CST, particularly in immunocompromised individuals [22, 25].
Empirical antibiotic therapy is aimed at broad-spectrum coverage, targeting the commonly associated pathogens in CST and frequently involves the intravenous use of nafcillin sodium/flucloxacillin, metronidazole, and ceftriaxone sodium. Thereafter, on-going antibiotic therapy should be appropriately modified upon the availability of culture results [22, 25]. Vancomycin is often substituted for nafcillin if there is a high suspicion for methicillin resistance [25]. Though there is no universal consensus on dosage and duration of antibiotic therapy, 3-4 weeks of high dose therapy appears to be a favoured recommendation contingent on the complete resolution of fever and clinical signs of inflammation during that period [22, 25]. Though surgical drainage of the CS is exceedingly rare, surgical intervention may be crucial in managing potential sources of infective thrombi such as primary sinusitis, oral infections, orbital abscesses and subdural empyemas [22, 25]. Early use of anti-coagulant therapy (within 7 days of hospitalisation for CST) may reduce mortality and morbidity rates, particularly in patients with unilateral disease and patients without cortical venous infarction. Though the choice of anticoagulant therapy is widely debated, heparin appears to be favoured with a recommended treatment duration of 4-6 weeks [22, 25]. Interestingly, the use of systemic corticosteroids to combat inflammation and oedema is not well-supported in patients with CST [25].
3.5. Vascular Lesions of the CS: ICA aneurysms and Carotid-Cavernous Fistulas (CCF)
Aneurysms originating from the cavernous portion (C4) of the intracranial ICA account for approximately 2%– 9% of all intracranial aneurysms and approximately 5% - 8% of all giant aneurysms (>2.5 cm) [3, 21, 26]. In adults, ICA cavernous segment aneurysms are the most common non-neoplastic lesions affecting the CS. Most ICA cavernous segment aneurysms are asymptomatic and are incidentally detected at imaging. CSS symptoms only tend to develop when the aneurysms become large (> 2.5 cm), more notably in females above the age of 50 [1, 3, 21]. The aetiology of ICA cavernous segment aneurysms can be classified as traumatic, infectious, or idiopathic. Though traumatic cerebral aneurysms are exceedingly rare, accounting for only 0.04-0.15% of all intracranial aneurysms, over 40% of them involve the cavernous and petrous segments of the ICA and are commonly associated with the base of skull fractures [27]. Infectious ICA cavernous segment aneurysms are a rare entity and are postulated to be a result of septic embolization precipitated by bacterial endocarditis [1, 27]. Risk factors for idiopathic ICA cavernous segment aneurysms include uncontrolled hypertension, smoking, familial genetic predisposition, and connective tissue disorders [27].
Like tumors involving the CS, clinical features are mainly mediated through the mass effect of the aneurysm, placing significant mechanical stress on intra-cavernous structures, particularly CN VI and the sympathetic plexus that are in close proximity [1-3, 26]. Extension of the aneurysm through the roof of the CS and subsequent rupture into the subarachnoid space can also result in symptoms consistent with subarachnoid hemorrhage such as severe headache, photophobia, and neck stiffness [2, 18]. Ophthalmic artery variants that originate from the cavernous portion of the ICA can further compound clinical presentation, as aneurysms can result in visual deficits or loss [28].
The risk of rupture for traumatic/idiopathic ICA cavernous segment aneurysms is relatively low and highly size-dependent, where 5-year rupture-risks for asymptomatic aneurysms measuring 1.3 – 2.4 cm and ≥2.5 cm are 3% and 6.4%, respectively. The rupture risk of infectious ICA cavernous segment aneurysms is not known [27]. The rupture of ICA aneurysms within the CS can lead to the rapid formation of direct carotid-cavernous fistulas (CCF) that are distinguished clinically by the classical quartet of chemosis, pulsatile proptosis, arterialized conjunctival veins and orbital bruit [1-4]. Patients also often develop the clinical spectrum of CSS, which includes symptoms consistent with impairment of the intracavernous cranial nerves and autonomic system. Indirect CCF can also occur via shunts stemming from the meningeal branches of the ICA supplying the CS dura. With these, CSS develops insidiously with patients presenting initially with only arterialized conjunctival veins, often without a bruit and pulsatile proptosis. Direct CCF tend to require complex surgical management, whereas indirect CCF tend to spontaneously resolve [2, 3, 27].
Treatment approaches for ICA cavernous segment aneurysms encompass constructive and deconstructive strategies which may or may not involve revascularization. Constructive strategies constitute direct microsurgical clipping, endovascular coil embolization (+/- utilization of intracranial stents or endoluminal balloons), liquid embolization and parent artery reconstruction without aneurysm embolization. Deconstructive strategies include the Hunterian method involving the occlusion of the ICA parent artery, often after a balloon test occlusion is performed to establish the need for revascularization [27].
3.6. Miscellaneous Non-infectious Inflammatory Disorders of the CS
The Tolosa-Hunt Syndrome (THS) is characterized by granulomatous inflammation of the anterior CS and the anteriorly related SOF and by its corticosteroid-responsive nature. Pathophysiology of THS remains undefined, and it is often a diagnosis of exclusion. Clinical presentation of THS tends to be sub-acute and often episodic with periods of spontaneous remissions. It commonly encompasses features of CSS such as retro-orbital pain, external and internal ophthal- moplegia [1, 3, 4]. Symptoms are largely due to the extensive formation of inflammatory granulomas and thrombus in the CS and its related venous channels and their resultant mass effect on cavernous structures such as the ICA and cavernous cranial nerves [3, 4, 21]. THS is postulated to be a part of a spectrum of inflammatory disorders which include inflammatory myo- fibroblastic tumors and idiopathic hypertrophic pachyme- ningitis, both of which have been noted to infiltrate the CS from their extracavernous origins and often mimic THS due to their histological and radiological similarities. THS responds well to high-dose corticosteroid therapy administered over a period of 3-6 months with subsequent slow weaning of therapy thereafter [21, 22]. Other systemic inflammatory disorders affecting the CS, though rare, include IgG4-related disease, granulomatosis with polyangiitis, sarcoidosis and tuberculosis [3, 21].
CONCLUSION
The CS is closely related to clinically significant neural and vascular structures, which are all susceptible to various neoplastic, infectious, vascular, and inflammatory disorders. As such, an astute understanding of the complex anatomy and pathology involving the CS will strengthen the diagnostic approaches in evaluating patients with CSS and make way for enhanced provision of relevant medical and surgical treatment.
CONSENT FOR PUBLICATION
Not applicable.
FUNDING
The authors declared that this review received no financial support.
CONFLICTS OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Declared none.


